 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
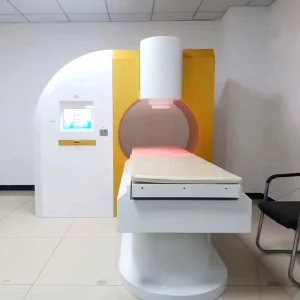 Tens + Thermal Stomach Therapy Device for Chronic Gastritis
Tens + Thermal Stomach Therapy Device for Chronic Gastritis Eswt Shockwave Therapy Device for Chronic Tendon Repair
Eswt Shockwave Therapy Device for Chronic Tendon Repair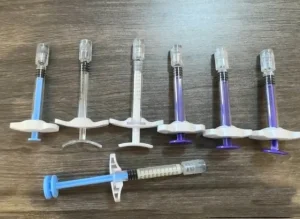 Triple Helix Collagen Structural Support Long Lasting Regenerative Scaffold Biomaterial
Triple Helix Collagen Structural Support Long Lasting Regenerative Scaffold Biomaterial Injectable Collagen Protein Human Collagen Scaffold Soft Tissue Augmentation Medical Use
Injectable Collagen Protein Human Collagen Scaffold Soft Tissue Augmentation Medical Use Advanced Pneumatic Shockwave Therapy Device for Chronic Pain Physiotherapy Treatment Centers
Advanced Pneumatic Shockwave Therapy Device for Chronic Pain Physiotherapy Treatment Centers Newangie Pmst Neo Magnetotherapy Device for Relief of Chronic Pain
Newangie Pmst Neo Magnetotherapy Device for Relief of Chronic Pain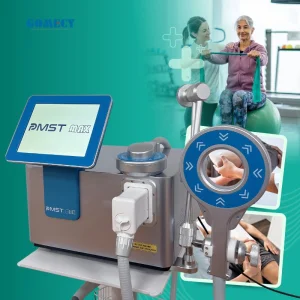 Electromagnetic Therapy Device for Chronic Pain Relief
Electromagnetic Therapy Device for Chronic Pain Relief Durable Ringlock Scaffold with Anti Corrosion and Anti Oxidation Protection
Durable Ringlock Scaffold with Anti Corrosion and Anti Oxidation ProtectionThe global Abdominal Aortic Aneurysm (AAA) repair market is witnessing a paradigm shift driven by technological maturity and an aging demographic. AAA, characterized by a localized dilation of the abdominal aorta, remains a critical cardiovascular challenge worldwide. Currently, the industry is dominated by Endovascular Aneurysm Repair (EVAR), a minimally invasive alternative to traditional open surgery. This segment alone accounts for over 70% of the total repair procedures in developed economies.
From a commercial perspective, the market is projected to reach several billion dollars by 2030, with North America and Europe leading in terms of surgical volume. However, the Asia-Pacific region is emerging as the fastest-growing hub, fueled by improved healthcare infrastructure and rising awareness. Factories specializing in vascular intervention must now navigate complex regulatory environments (MDR in Europe, FDA in the US, NMPA in China) while maintaining rapid innovation cycles.
Development of ultra-low profile stent grafts that can navigate through highly tortuous and narrow iliac arteries, expanding the eligibility for EVAR.
Artificial Intelligence is being integrated into pre-operative CT analysis to automatically size and select the optimal graft configuration for each patient.
Research into scaffolds that provide temporary support and then slowly dissolve, allowing the vessel to regain its physiological elasticity.
Enhanced imaging software allowing top-tier surgeons to consult or even assist in robotic-assisted AAA repairs remotely across the globe.
Factories are moving towards patient-specific grafts created via 3D printing to handle complex fenestrated or branched aneurysm cases.
Innovations in polymer coatings and sealing rings to reduce the risk of Type II endoleaks, the most common complication post-EVAR.

Hangzhou SJ Medical Co., Ltd., established in 2015, stands at the forefront of the vascular interventional medical device industry. Our core mission is the research, development, and commercialization of high-end medical devices specializing in cardiovascular and peripheral intervention.
Our commitment to E-E-A-T (Experience, Expertise, Authoritativeness, and Trustworthiness) is reflected in our robust R&D pipeline. We don't just manufacture; we innovate. Our product portfolio includes precision balloon catheters, advanced stent systems, and diagnostic access devices designed to meet the rigorous demands of modern surgery. By holding multiple patents and international certifications, we provide a foundation of reliability for healthcare providers in over 50 countries.
SJ Medical operates a state-of-the-art 5,000 square meter facility. Built strictly to GMP standards, our factory features a million-class clean workshop, with localized hundred-class areas for critical high-precision processes. This environment ensures that every product, from a simple guide wire to a complex balloon catheter, is free from contamination and consistent in quality.
Under the guidance of industry veterans and senior engineers, our technical team maintains a culture of continuous improvement. We utilize high-standard production equipment, including laser welding, marking technologies, and precision balloon forming systems to ensure surgical-grade accuracy.
Global procurement teams in the medical sector are increasingly looking to China, and specifically to hubs like Hangzhou, for their supply chain needs. The reasons extend beyond cost-effectiveness:
| Feature | SJ Medical (China) | Traditional Western Exporters | Impact on Buyers |
|---|---|---|---|
| R&D Cycle | 6-12 Months | 18-24 Months | Faster access to innovation |
| Customization | High Flexibility (OEM/ODM) | Limited / Rigid | Tailored to local clinical needs |
| Production Cost | Highly Competitive | Premium Pricing | Better hospital margins |
| Quality Control | Certified (CE/ISO/GMP) | Certified (FDA/CE) | Equal safety standards |
Abdominal Aortic Aneurysm repair products are not "one size fits all." Different regions have unique clinical needs. For instance, in European markets (Italy, France, Spain), there is a high demand for low-profile grafts due to the aging population's vascular morphology. In Emerging Markets (India, Indonesia, Egypt), the focus is often on durability and cost-effective access systems that can perform reliably in varied hospital settings.
SJ Medical has successfully established a global network. Our products are trusted in Finland, Italy, Spain, the Netherlands, and across the Middle East. We provide comprehensive services from pre-sales technical consultation to responsive after-sales support, ensuring that clinicians are never alone in the operating theater.

 Colorful Foldable Galvanized Scaffold for Customized Renovation Projects
Colorful Foldable Galvanized Scaffold for Customized Renovation Projects Quick Deploy Folding Scaffold for Changing Sites and Efficient Operations
Quick Deploy Folding Scaffold for Changing Sites and Efficient Operations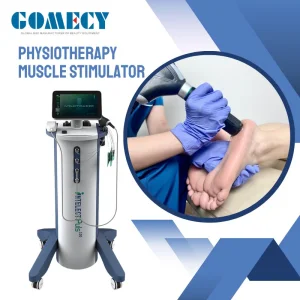 Medical Grade Electrotherapy Device for Chronic Pain Relief Compact Electrical Stimulation Device
Medical Grade Electrotherapy Device for Chronic Pain Relief Compact Electrical Stimulation Device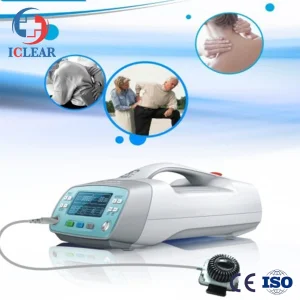 Ce 500MW Portable Home Use Medical Semiconductor Pain Relief Laser Therapy Device for Chronic Athletic Pain
Ce 500MW Portable Home Use Medical Semiconductor Pain Relief Laser Therapy Device for Chronic Athletic Pain Poly Lactic Acid Polymer for Tissue Engineering Scaffolds Biodegradable
Poly Lactic Acid Polymer for Tissue Engineering Scaffolds Biodegradable Lifting Medical Grade PLA for Sustained Release Injectables Tissue Engineering Scaffolds
Lifting Medical Grade PLA for Sustained Release Injectables Tissue Engineering Scaffolds Biodegradable Plga Microspheres for Drug Delivery Surgical Sutures Tissue Engineering Scaffolds
Biodegradable Plga Microspheres for Drug Delivery Surgical Sutures Tissue Engineering Scaffolds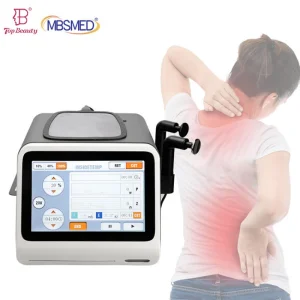 Portable 448kHz Indiba Tecar Therapy Device for Anti-Inflammation & Chronic Pain Relief
Portable 448kHz Indiba Tecar Therapy Device for Anti-Inflammation & Chronic Pain Relief