 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
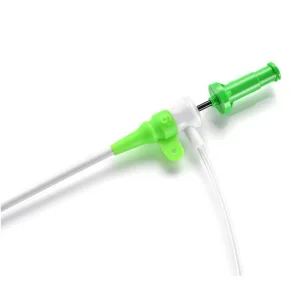
Disposable Products 4f-10f Radial Vascular Guide Wire Introduction Catheter Kit
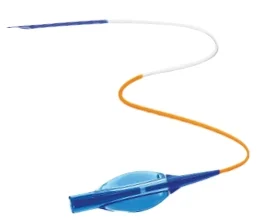
Advanced Thrombus Extraction Catheter for Efficient Blood Clot Removal
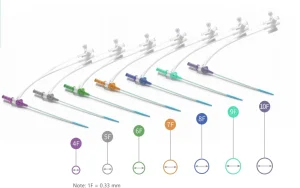
Microapproach CE Disposable Femoral Radial Vascular Access Introducer Sheath Kit
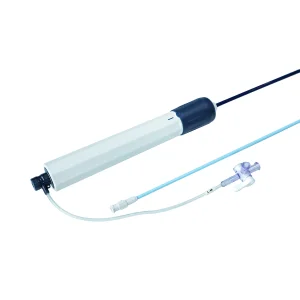
High Quality Intracardiac Access Sheath for Radiofrequency Ablation Patient



Muscle Relax Leg Recovery Boot Air Compression Therapy System
The global molecular diagnostics market has witnessed an unprecedented transformation over the last few years. As the cornerstone of genetic testing, the Viral DNA RNA Extraction Kit industry has transitioned from a specialized research tool to a high-demand clinical necessity. In the post-pandemic era, the emphasis on early detection of infectious diseases—ranging from respiratory viruses to blood-borne pathogens—has made high-quality extraction kits vital for public health infrastructure.
Currently, the market is moving toward high-throughput and automated solutions. Traditional spin-column methods, while still reliable, are being rapidly supplemented or replaced by magnetic bead-based technology. This shift is driven by the need for speed, consistency, and the integration of artificial intelligence in lab workflows. As a leading exporter, we recognize that the quality of nucleic acid purification directly impacts downstream applications like PCR, NGS (Next-Generation Sequencing), and gene expression analysis.
Labs are minimizing human error by adopting fully automated workstations. Our kits are optimized for compatibility with major robotic platforms, ensuring seamless integration into modern laboratory ecosystems.
Advanced surface chemistry on superparamagnetic beads allows for higher binding capacity and faster elution times, crucial for rapid viral load monitoring and emergency diagnostics.
Point-of-Care Testing (POCT) requires simplified extraction steps. We are developing "extraction-free" or "one-step" lysis buffers that maintain high sensitivity for field use.
Reducing plastic waste and utilizing biodegradable components in kit packaging is becoming a standard for global tenders and environmentally conscious healthcare systems.
Global procurement teams from hospitals, CDC (Center for Disease Control), and private diagnostic laboratories look for more than just a product; they seek a reliable supply chain partner. The demand for Viral DNA RNA Extraction Kits is no longer localized. From the intensive research facilities in Europe to the burgeoning clinical labs in Southeast Asia and the Middle East, the criteria for selection remain stringent: Lot-to-lot consistency, shelf-life stability, and comprehensive technical documentation.
As a specialized manufacturer, we address localized application scenarios. For instance, in regions with extreme climates, our kits are formulated for thermal stability during transport. In high-volume screening scenarios, our 96-well plate formats are designed to maximize throughput without compromising the integrity of the viral RNA.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. While our expertise lies deep in the cardiovascular and peripheral intervention fields, our commitment to sterile manufacturing and precision engineering has allowed us to excel as a trusted partner in the broader medical device and diagnostic landscape.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
Under the leadership of senior engineers and a technical team with decades of experience, SJ Medical maintains strict quality control and continuously improves its manufacturing processes to ensure product reliability and consistency across all lines, including interventional access systems and diagnostic tools.
Our proximity to the world's most robust electronics and chemical supply chains allows us to prototype and refine extraction formulas faster than competitors in the West.
Scale of economy enables us to offer premium, CE-certified extraction kits at a price point that makes mass screening and longitudinal research economically viable.
Adhering to ISO 13485 and CE standards, our factory ensures that every batch undergoes stringent QC, from raw material inspection to final functional testing.
Successful viral detection starts with the Lysis stage. Our proprietary lysis buffers are engineered to efficiently disrupt viral envelopes and capsids, even in complex samples like whole blood, sputum, or environmental swabs, while simultaneously inactivating RNases to protect the fragile RNA from degradation.
During the Binding phase, our silica-coated magnetic beads exhibit a high surface-area-to-volume ratio, ensuring maximum capture of nucleic acids. The Washing steps effectively remove proteins, salts, and other PCR inhibitors, resulting in ultra-pure DNA/RNA elution. This high level of purity is essential for high-sensitivity diagnostics, where "false negatives" are not an option.
Whether you are an OEM partner looking for bulk reagents or a clinical distributor needing branded kits, our facility in Hangzhou is equipped to meet your specific technical requirements with precision and authority.
SJ Medical strictly adheres to international quality standards. The company has obtained CE certification, ISO 13485 quality management system certification, and CFDA approval, along with free sale certificates. These credentials demonstrate our strong commitment to product quality and compliance on a global scale.
We have established long-term partnerships with clients across Europe, Asia, the Middle East, and Africa. Our products are currently distributed in countries such as Italy, Spain, France, the Netherlands, India, Indonesia, Turkey, and Egypt. We continue to strengthen our global presence through innovation and responsive after-sales service.

Absorbable and Nonabsorbable Sutures: Strength and Bioresorption
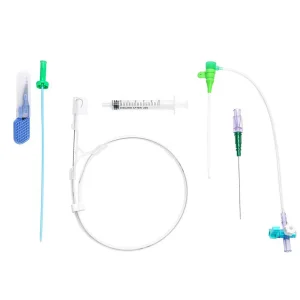
Premium Vascular Access Catheter Sheath for Reliable Hospital Use

Premium Vascular Access Introducer Catheter Sheath for Hospitals
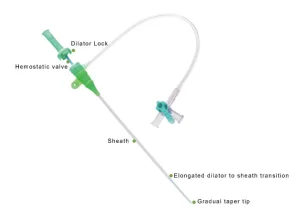
Premium Disposable Catheter Sheath for Enhanced Vascular Access

Premium Vascular Access Catheter Sheath for Medical Professionals

Premium Medical Introducer Catheter Sheath for Enhanced Performance
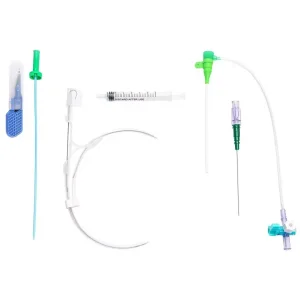
Angiography Catheter Sheath - Disposable Medical Supply Solutions
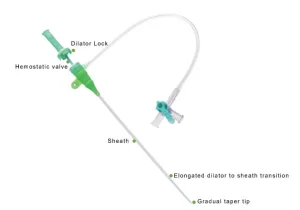
Premium Versatile Angiography Catheter Sheath - Multiple Sizes
From pre-sales consultation and technical support to efficient order processing and responsive after-sales service, SJ Medical ensures that every customer receives timely assistance and reliable solutions. Our expertise in vascular intervention and molecular diagnostic manufacturing makes us the premier choice for global exporters and medical institutions.
