 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
 Terahertz Wave Foot Massage Device with Pemf for Chronic Pain Relief Olylife
Terahertz Wave Foot Massage Device with Pemf for Chronic Pain Relief Olylife
 FDA Ce 6 Chamber Air Compression Lymphatic Drainage Device for People Who Has Chronic Venous Insufficiency
FDA Ce 6 Chamber Air Compression Lymphatic Drainage Device for People Who Has Chronic Venous Insufficiency
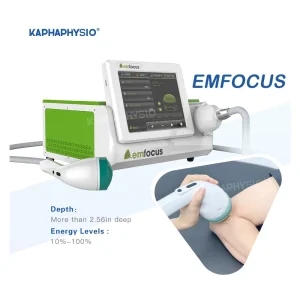 Professional Shock Wave Medical Device for Chronic Pain Management
Professional Shock Wave Medical Device for Chronic Pain Management
 Advanced Optical Therapy Device for Chronic Constipation and Osteoarthritis
Advanced Optical Therapy Device for Chronic Constipation and Osteoarthritis
 Eswt Shockwave Therapy Device for Chronic Tendon Repair
Eswt Shockwave Therapy Device for Chronic Tendon Repair
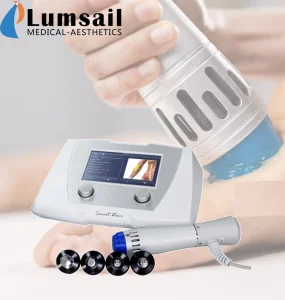 Radial/Linear Shockwave Device for Chronic Myofascial Pain Syndrome
Radial/Linear Shockwave Device for Chronic Myofascial Pain Syndrome
 Health Care Electric Potential Therapy Device for Chronic Pain and Insomnia
Health Care Electric Potential Therapy Device for Chronic Pain and Insomnia
 Advanced Pneumatic Shockwave Therapy Device for Chronic Pain Physiotherapy Treatment Centers
Advanced Pneumatic Shockwave Therapy Device for Chronic Pain Physiotherapy Treatment Centers
The global medical community defines Chronic Total Occlusion (CTO) as a complete or near-complete blockage of a coronary or peripheral artery that has been present for at least three months. Historically, CTOs were considered the "final frontier" of interventional cardiology due to their high procedural complexity and lower success rates compared to non-occlusive lesions. However, the industrial landscape has shifted dramatically.
The global CTO devices market is projected to grow at a CAGR of over 7.5% through 2030, driven by an aging population and the rising prevalence of Coronary Artery Disease (CAD).
Manufacturing CTO devices requires extreme precision. Microcatheters and guidewires must balance "pushability," "torqueability," and "trackability" to navigate calcified lesions.
Modern CTO procedures utilize advanced techniques like Antegrade Wire Escalation (AWE) and Retrograde approaches, demanding specialized equipment for each step.
As a leading manufacturer, we observe several key trends shaping the future of CTO interventions:
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.

Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.
SJ Medical operates a modern manufacturing facility built in accordance with GMP standards. Our facility features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
Equipped with precision balloon forming equipment, laser welding, and marking technologies to ensure high levels of manufacturing accuracy.
Our technical team consists of industry veterans with decades of experience in medical device engineering and clinical application.
Full-process quality control from raw material inspection to final sterilization, ensuring product reliability and consistency.
As a premier CTO devices exporter, we understand the diverse needs of global procurement officers and clinicians. The demand for high-quality interventional tools is not uniform; it varies by region and clinical setting.
The shift toward Chinese medical device manufacturing is driven by the "Value-Based Healthcare" model. Companies like SJ Medical offer:
SJ Medical focuses on the development of vascular interventional devices designed for both coronary and peripheral procedures. Each product is developed with an emphasis on precision, safety, and clinical performance.
PTCA Balloon Catheters, CTO Guidewires, and Microcatheters designed for complex lesion crossing.
PTA Balloon Catheters and access systems for treating Peripheral Artery Disease (PAD).
Diagnostic catheters and introducer sheaths that provide the foundation for successful procedures.
Our products are currently distributed in countries such as Finland, Italy, Spain, France, the Netherlands, India, Indonesia, Turkey, and Egypt. We continue to expand our international footprint to support physicians in achieving optimal procedural outcomes.

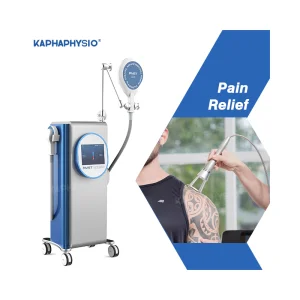 Advanced Magnetic Therapy Device for Chronic Pain Relief
Advanced Magnetic Therapy Device for Chronic Pain Relief
 Manufacturer Offer High Electric Potential Therapy Device to Treat Insomnia Headache Chronic
Manufacturer Offer High Electric Potential Therapy Device to Treat Insomnia Headache Chronic
 Newangie Pmst Neo Magnetotherapy Device for Relief of Chronic Pain
Newangie Pmst Neo Magnetotherapy Device for Relief of Chronic Pain
 Portable Handheld Red Light Therapy Device with Pulsed Settings for Effective Acute and Chronic Pain Therapy
Portable Handheld Red Light Therapy Device with Pulsed Settings for Effective Acute and Chronic Pain Therapy
 Terahertz Foot Massage Device Pemf for Chronic Pain Relief Olylife Thz P90
Terahertz Foot Massage Device Pemf for Chronic Pain Relief Olylife Thz P90
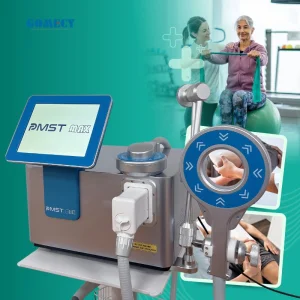 Electromagnetic Therapy Device for Chronic Pain Relief
Electromagnetic Therapy Device for Chronic Pain Relief
 Low Level Laser Therapy Device for Chronic Acute Joint Pain
Low Level Laser Therapy Device for Chronic Acute Joint Pain
 Portable 448kHz Indiba Tecar Therapy Device for Anti-Inflammation & Chronic Pain Relief
Portable 448kHz Indiba Tecar Therapy Device for Anti-Inflammation & Chronic Pain Relief