 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
The medical landscape in South Africa is undergoing a significant transformation, particularly in the field of interventional cardiology. As a leading Bioresorbable Vascular Scaffolds exporter, we recognize that the South African healthcare system, characterized by a sophisticated private sector and a broad public sector, is increasingly demanding high-tech solutions to combat cardiovascular diseases (CVD). With the burden of heart-related ailments rising in urban hubs like Johannesburg, Cape Town, and Durban, the shift from traditional metallic Drug-Eluting Stents (DES) to Bioresorbable Vascular Scaffolds (BVS) represents a monumental leap in patient care.
South Africa's cardiology market is unique. The South African Health Products Regulatory Authority (SAHPRA) maintains stringent quality controls, ensuring that only medical devices with proven efficacy and safety enter the market. Our exports to South Africa focus on providing scaffolds that naturally dissolve over time, leaving no permanent metal "cage" in the artery. This "vascular restoration therapy" is becoming the preferred choice for younger South African patients who wish to avoid the long-term risks associated with permanent implants, such as late-stent thrombosis.
Our scaffolds facilitate the restoration of vasomotor function, allowing the coronary artery to respond naturally to physiological demands—a critical factor for the active South African lifestyle.
With South Africa serving as a hub for the SADC region, our export services ensure that the latest Sirolimus-eluting technology reaches clinics from Sandton to Pretoria with full cold-chain integrity.
Leveraging decades of R&D expertise, we provide South African cardiologists with technical documentation, clinical trial data, and localized support that meets global standards.
Hangzhou SJ Medical Co., Ltd., established in 2015, stands at the forefront of vascular intervention technology. We are not just an exporter; we are innovators dedicated to the research, development, and manufacturing of high-end medical devices. Our focus spans both cardiovascular and peripheral interventions, providing safe and reliable solutions to healthcare providers in South Africa and beyond.
Our commitment to the South African market is rooted in Expertise and Authoritativeness. Our technical team comprises senior engineers with decades of experience in the biocompatible polymer industry. By focusing on materials like Poly Lactic Acid (PLA) and advanced collagen structures, we ensure that our scaffolds offer the mechanical strength of metal during the healing phase, while being fully resorbable thereafter.

The South African cardiology industry is witnessing a move towards "leave nothing behind" strategies. Modern surgeons at top-tier facilities like the Chris Hani Baragwanath Academic Hospital and various Netcare heart centers are prioritizing long-term vessel health. The key trends driving the adoption of Bioresorbable Vascular Scaffolds include:
As a specialized exporter, SJ Medical ensures that our Sirolimus-Eluting Bioresorbable Coronary Stent Implants are supplied with full regulatory compliance and clinical training materials for South African practitioners. Our production facility in Hangzhou, spanning 5,000 square meters and built to GMP standards, serves as the engine for this global supply chain.
Our manufacturing prowess is anchored in a million-class clean workshop with localized hundred-class areas for critical assembly processes. This environment is essential for producing bioresorbable devices that are ultra-sensitive to contaminants. We utilize precision balloon forming equipment and laser welding technology to ensure that every scaffold exported to South Africa meets the highest "Reliability" threshold of the E-E-A-T principle.
Certifications: We are proud holders of CE certification, ISO 13485 quality management system certification, and CFDA approval. Our Free Sale Certificates allow for seamless entry into the South African medical distribution network.

Our Bioresorbable Vascular Scaffolds are designed for diverse clinical applications within the South African demographic:
Treating congenital heart conditions or early-onset coronary artery disease in younger patients without permanent implants that might limit growth or future interventions.
Providing structural support for complex bifurcations in peripheral vascular diseases, common in diabetic patients across South Africa.
Meeting the expectations of private healthcare patients in Sandton or Umhlanga who demand the most advanced, minimally invasive technology available globally.
 Safe Durable Multidirectional Scaffold (CE SGS Approved)
Safe Durable Multidirectional Scaffold (CE SGS Approved)
 South Africa Clinical Sutures: Strength & Bioresorption
South Africa Clinical Sutures: Strength & Bioresorption
 Modern Multidirectional Scaffolds - Safety Guaranteed
Modern Multidirectional Scaffolds - Safety Guaranteed
 South Africa Specialized Tube & Coupler Scaffolds
South Africa Specialized Tube & Coupler Scaffolds
 HADM Long-Acting Collagen Medical Device South Africa
HADM Long-Acting Collagen Medical Device South Africa
 Versatile Electrophoresis Steel Scaffold for SA Projects
Versatile Electrophoresis Steel Scaffold for SA Projects
 Biobase Pathological Tissue Embedding Center
Biobase Pathological Tissue Embedding Center
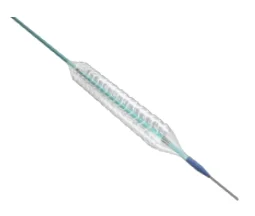 Sirolimus-Eluting Bioresorbable Coronary Stent South Africa
Sirolimus-Eluting Bioresorbable Coronary Stent South Africa
SJ Medical has established a robust distribution network spanning Europe, Asia, the Middle East, and notably, Africa. In South Africa, our role as a primary Bioresorbable Vascular Scaffolds exporter is supported by local partnerships that understand the intricacies of the South African medical procurement process. We supply not only the physical scaffold but also the specialized delivery systems (balloon catheters) and diagnostic tools required for successful implantation.
Our research indicates that the South African market for interventional cardiology is poised for a CAGR of over 6% in the next five years. This growth is driven by increasing healthcare expenditure and a growing preference for minimally invasive surgeries. As exporters in South Africa, we are committed to closing the gap between cutting-edge global technology and local patient access. Every product we ship is backed by comprehensive technical support, ensuring that clinicians in Cape Town or Johannesburg have the same level of confidence as those in London or Milan.
Navigating the regulatory environment of South Africa requires a partner with deep Experience and Trustworthiness. SJ Medical’s adherence to ISO 13485 and our proactive engagement with international regulatory bodies ensure that our products meet the "Safety First" ethos of the South African medical community. Our lab facilities conduct rigorous testing on:
Whether you are a medical device distributor in Gauteng or a hospital procurement officer in the Western Cape, SJ Medical provides the authoritative partnership needed to elevate vascular care in South Africa. We invite you to explore our range of Bioresorbable Vascular Scaffolds and experience the difference that precision engineering and professional export management can make for your clinical outcomes.