 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
New Zealand's medical technology (MedTech) sector is witnessing a paradigm shift, particularly in the realm of interventional cardiology. As the demand for minimally invasive procedures grows within the New Zealand District Health Boards (DHBs), the focus has intensified on Bioresorbable Vascular Scaffolds (BVS). Unlike traditional permanent metallic stents, BVS technology provides temporary mechanical support to the vessel wall before being fully absorbed by the body. This "disappearing" act restores the natural vasomotion of the artery, a critical factor for long-term vascular health in patients across Auckland, Wellington, and Christchurch.
The industrial landscape in New Zealand is uniquely positioned. With a strong emphasis on R&D through institutions like the Auckland Bioengineering Institute, the country has become a testing ground for advanced biomaterials. Factories and exporters focusing on Bioresorbable Vascular Scaffolds in New Zealand are leveraging high-grade polymers such as Poly-L-Lactic Acid (PLLA). These materials are chosen for their excellent biocompatibility and predictable degradation rates, ensuring that the scaffold maintains its integrity during the crucial 6-month healing period before undergoing hydrolysis.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
The company is supported by a highly experienced technical team, many of whom have been engaged in the medical device industry for decades. Under the leadership of senior engineers, SJ Medical maintains strict quality control and continuously improves its manufacturing processes to ensure product reliability and consistency.
Our vascular interventional devices are engineered for precision. From diagnostic catheters to therapeutic sirolimus-eluting stents, each component undergoes rigorous testing to meet the demands of New Zealand's rigorous medical standards.
Specialized scaffolds for treating coronary artery disease, ensuring luminal patency while facilitating vascular remodeling.
Solutions for lower limb arterial disease, designed to withstand the dynamic mechanical stress of peripheral vessels.
SJ Medical strictly adheres to international quality standards and regulatory requirements. The company has obtained CE certification, ISO 13485 quality management system certification, and CFDA approval, along with free sale certificates, demonstrating its strong commitment to product quality and compliance.

 Safe Durable Multidirectional Scaffold with CE SGS Approved for NZ Markets
Safe Durable Multidirectional Scaffold with CE SGS Approved for NZ Markets
 Medical Grade Absorbable and Nonabsorbable Sutures: Combining Strength and Bioresorption
Medical Grade Absorbable and Nonabsorbable Sutures: Combining Strength and Bioresorption
 Safety Guaranteed Modern Multidirectional Scaffolding System
Safety Guaranteed Modern Multidirectional Scaffolding System
 Complex Structure Scaffold Prop and Coupler Systems
Complex Structure Scaffold Prop and Coupler Systems
 Hadm Long-Acting Collagen Biological Scaffold Medical Device
Hadm Long-Acting Collagen Biological Scaffold Medical Device
 Versatile Electrophoresis Steel Scaffold for Global Infrastructure
Versatile Electrophoresis Steel Scaffold for Global Infrastructure
 Biobase Pathological Tissue Embedding Center for NZ Laboratories
Biobase Pathological Tissue Embedding Center for NZ Laboratories
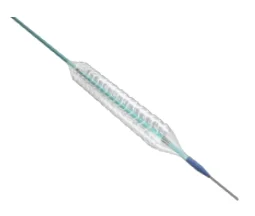 Advanced Sirolimus-Eluting Bioresorbable Coronary Stent Implant
Advanced Sirolimus-Eluting Bioresorbable Coronary Stent Implant
 Durable Ringlock Scaffold with Anti Corrosion and Anti Oxidation Protection
Durable Ringlock Scaffold with Anti Corrosion and Anti Oxidation Protection
 Vertical Standard Scaffold for Engineering Projects
Vertical Standard Scaffold for Engineering Projects
 High Mobility Scaffold for New Zealand Installation Projects
High Mobility Scaffold for New Zealand Installation Projects
 Durable Steel Flexible Cantilever Scaffold System
Durable Steel Flexible Cantilever Scaffold System
Join hands with a leading global exporter of vascular interventional devices. Whether you are in Auckland, Wellington, or abroad, our team is ready to provide technical excellence and reliable support.
Contact Our NZ Export Team