

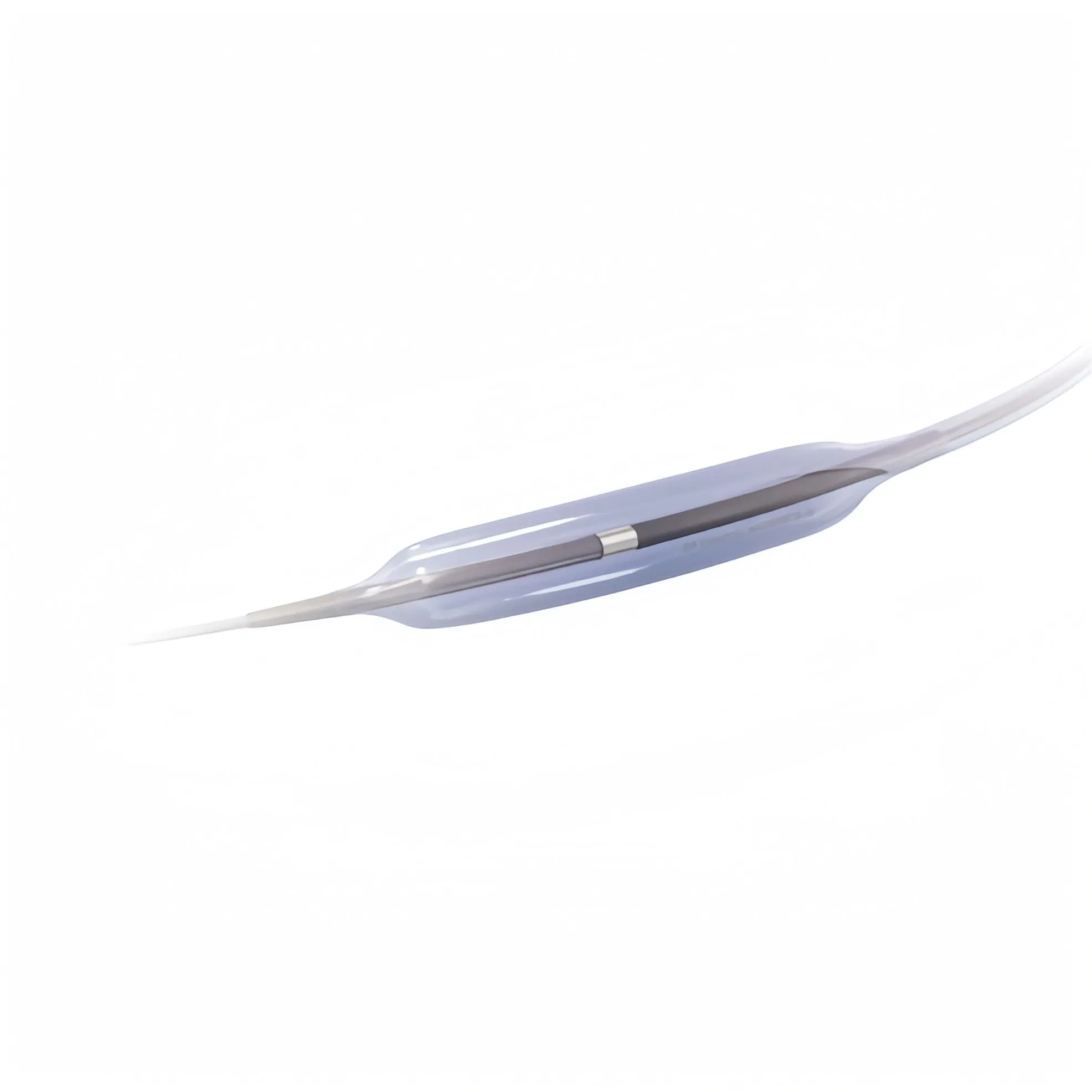


1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| Material: | Nylon, Pebax |
| Sterilization Method: | Ethylene Oxide Sterilization |

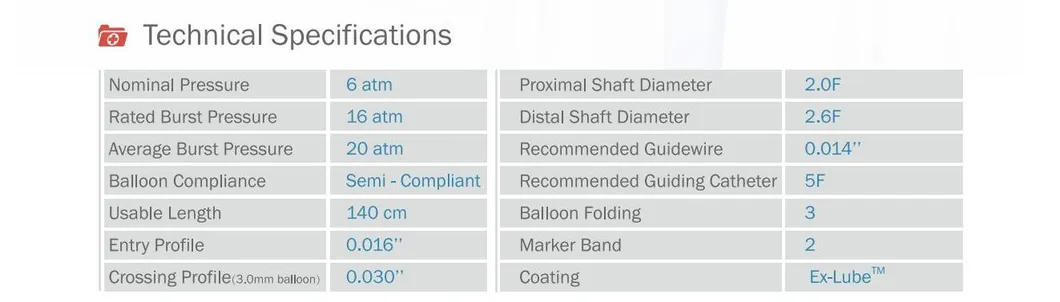
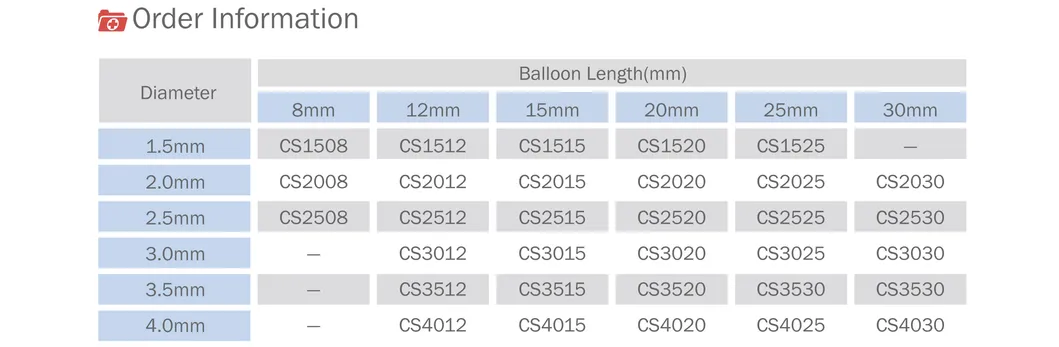

Founded in 2016, we specialize in coronary interventional medical devices and core parts. We welcome OEM/ODM cooperation and provide professional manufacturing services.
Our original PTFE coating and tube making technology originated from Japan, utilized in the medical device field for more than 30 years. Our team consists of experienced experts dedicated to high-quality production.
With 7,000 square meters of professional facilities and laboratories, we have obtained more than 50 intellectual property rights and patents, passing ISO 9001 and ISO 13485 quality systems.


Our products are recognized for great quality and competitive pricing in both domestic and overseas markets.


