


1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| OEM: | Accept OEM |
| Tolerance: | +/-0.02mm |
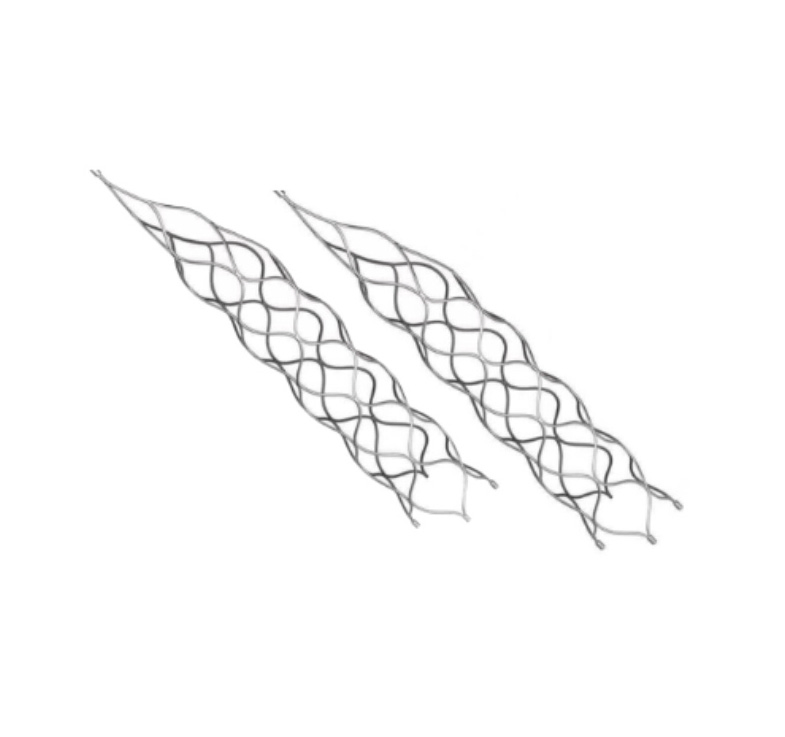
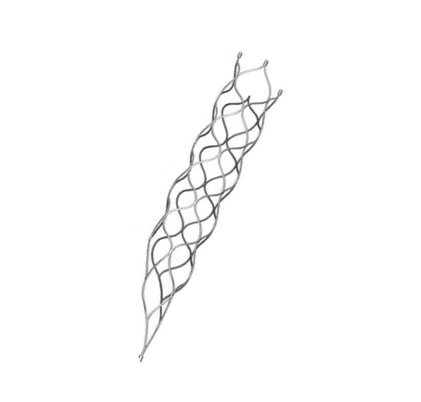
This product is a professional medical device specifically engineered for the treatment of vascular diseases. The primary material construction utilizes high-grade nickel-titanium tubing, ensuring durability and biological compatibility.
Application Scope: This medical device is designed for various surgical applications within the vascular specialty.

Established in 2019, our facility is a comprehensive center integrating medical device research, development, production, and sales services.








