



1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| Coating: | Teflon & Hydrophilic Coating |
| Tolerance: | +/-0.005mm |
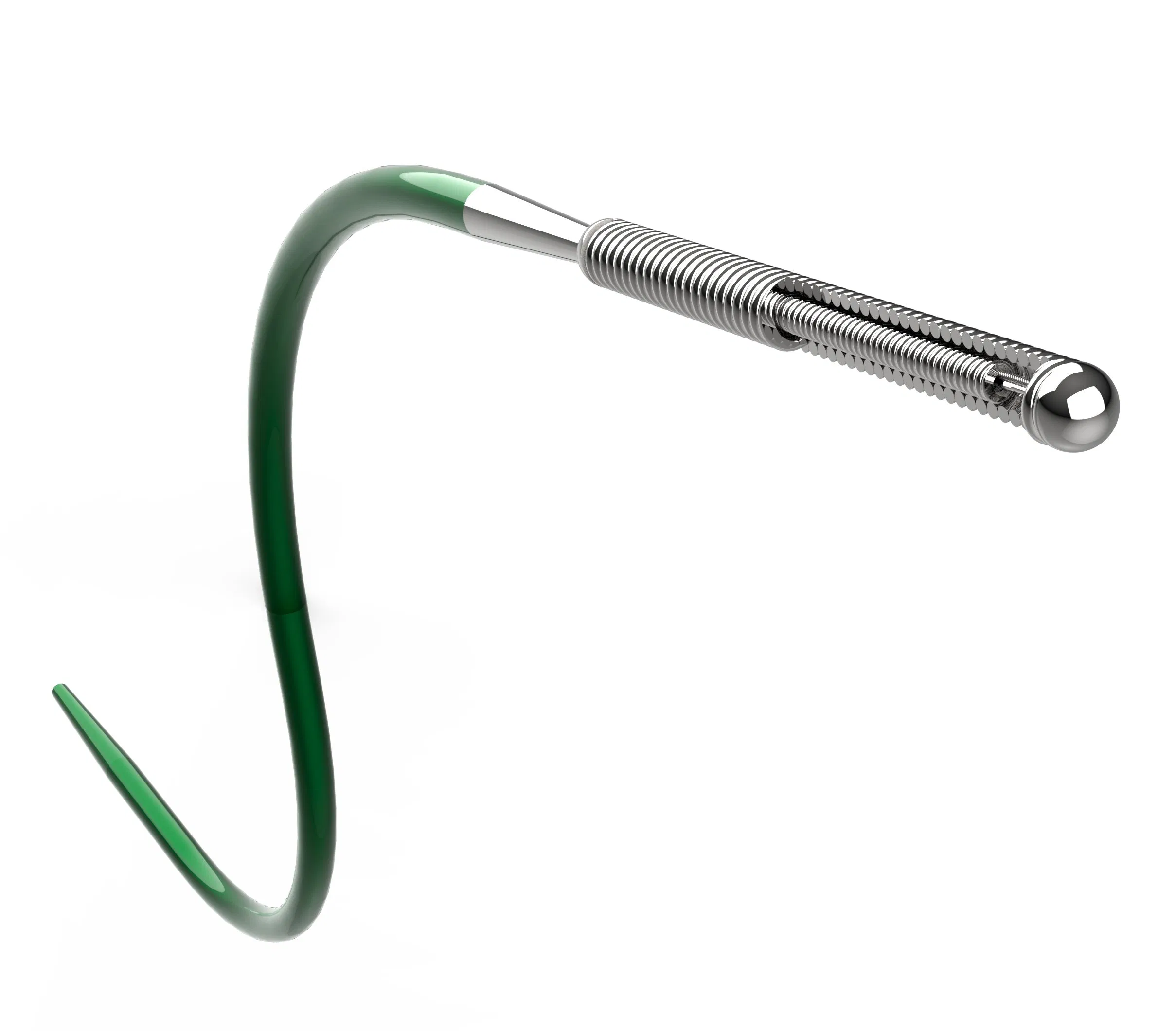
| Component | Material & Function |
|---|---|
| Inner Core Wire | Superelastic Nitinol (SE508), Ø0.005"-0.008", provides shape recovery and kink resistance |
| Outer Support Core | High-yield stainless steel (316LVM), Ø0.010"-0.012", enhances pushability and torque transmission |
| Taper Transition | Laser gradient welding (Tensile strength ≥80gf at transition zone) |
| Parameter | Dual-Core | Single-Core |
|---|---|---|
| Push Efficiency | +40% improvement (≥92% transmission) | 65-75% typical |
| Kink Resistance | >500 cycles (1.5mm bend radius) | 200-300 cycles |
| Tip Responsiveness | <5° torque delay (1:1.05 ratio) | 10-15° delay |

Complex Lesions:
Compatibility:
Established in 2019, we are a comprehensive entity integrating medical device R&D, production, and service. The facility has passed ISO13485:2016 certification, covering over 5,000 square meters with a professional workforce of over 100 people.









