



1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| Type: | Surgical Supplies Materials |
| Material: | PU |

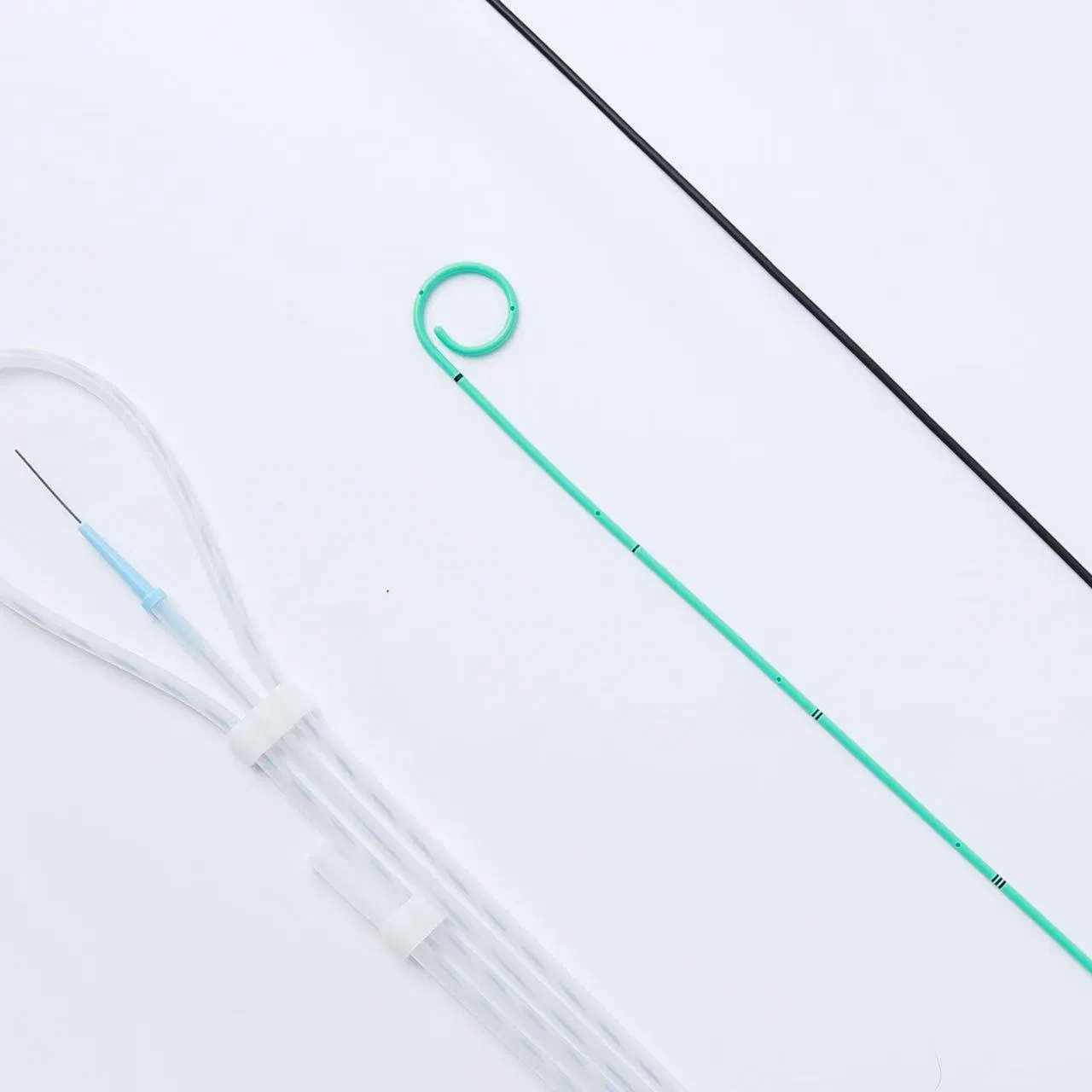
The Disposable Ureteral Stent consists of a basic configuration of a double-J tube and an optional configuration of a guide wire and a booster tube.
Made of premium polyurethane material, ensuring excellent biocompatibility and minimizing the risk of allergic reactions.
The stent is fully radiopaque under X-ray imaging, providing enhanced visibility to assist physicians in accurately confirming placement.
| Feature | Details |
|---|---|
| Product Name | Disposable Ureteral Stent |
| Sterilization | Ethylene Oxide Sterilization |
| Length | 100mm - 300mm |
| Specification | F3, F4, F4.5, F4.8, F5, F6, F7, F8 |
| Combination Package | Ureteral Stent, Guidewire, Boosting Tube |
| Instrument Classification | Class II |
| Term of Validity | 3 Years |
| Packing | Plastic Package |






Range of Application: This product is intended for stenting and drainage of the human ureter.