📝
Product Description
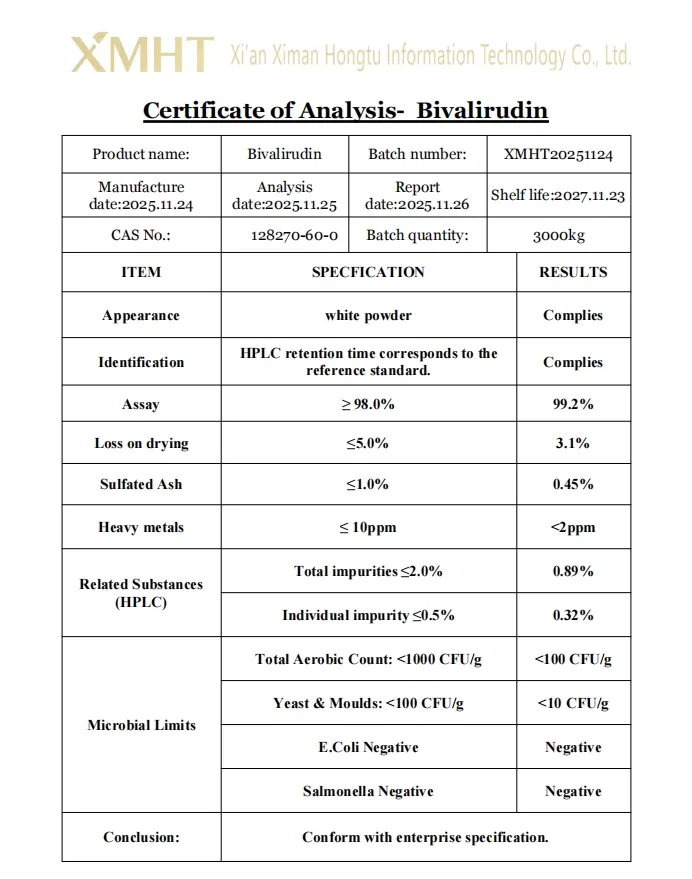
Bivalirudin is a synthetic 20 amino acid peptide direct thrombin inhibitor presented as a white to off-white lyophilized powder. It is highly soluble in water and in physiological saline solutions. The peptide sequence is H-D-Phe-Pro-Arg-Pro-Gly-Gly-Gly-Gly-Asn-Gly-Asp-Phe-Glu-Glu-Ile-Pro-Glu-Glu-Tyr-Leu-OH with a molecular weight of approximately 2180 Daltons. Identity is confirmed by methods such as HPLC and by peptide mapping using mass spectrometry. Purity is determined by reversed-phase HPLC, typically not less than 98.0 percent. Water content by Karl Fischer titration is controlled, usually not more than 8.0 percent. Bacterial endotoxins are limited to not more than 100 EU per milligram for the final drug product.
⚙️
Application & Function
Bivalirudin functions as a direct and reversible inhibitor of thrombin, a key enzyme in the blood coagulation cascade. It specifically binds to both the active catalytic site and the substrate recognition exosite 1 of thrombin, effectively blocking its ability to convert fibrinogen to fibrin and to activate platelets. It is indicated as an anticoagulant in adult patients undergoing percutaneous coronary intervention (PCI), including those with unstable angina or undergoing stent placement. It offers the advantage of a short half-life (approximately 25 minutes) and its effect diminishes rapidly after discontinuation, which may reduce bleeding risks compared to other anticoagulants.
❓
Frequently Asked Questions
What is Bivalirudin used for?
It is primarily used as an anticoagulant in patients undergoing percutaneous coronary intervention (PCI) and as an alternative to heparin.
What is the purity of your Bivalirudin powder?
Our Bivalirudin powder has a purity level of over 99%, tested by HPLC methods.
How should the product be stored?
The lyophilized powder should be stored at 2 to 25 degrees Celsius. Reconstituted solutions should be used immediately.
What is the advantage of Bivalirudin over heparin?
Bivalirudin has a shorter half-life (approx. 25 mins) and offers a more predictable anticoagulant effect with a lower risk of major bleeding in certain populations.
Is this product pharmaceutical grade?
Yes, it is Pharmaceutical Grade and complies with GMP, USP, and BP standards.
What is the physical state of the product?
The product is provided as a white to off-white lyophilized powder which is highly soluble in water.





 SJ Medical
SJ Medical