SJ Medical
SJ Medical










| Still deciding? Get samples of $ ! US$ 39.6/Piece Request Sample |
The sheath provides ureteral dilation and a continuous working tract for the introduction of endoscopes and instruments during ureteral access procedures. It protects the ureter during repeated instrument exchanges and shields the endoscope from damage. Featuring a hydrophilic coating, the surface becomes lubricious, significantly reducing friction and potential damage to the ureteral wall.
Structure & CompositionThe product consists of a guide sheath and a dilator. The guide sheath includes a hydrophilic-coated catheter and a joint. The dilator consists of a dilator catheter, joint, and chuck. The tip of the dilator is also hydrophilic-coated for smooth insertion.
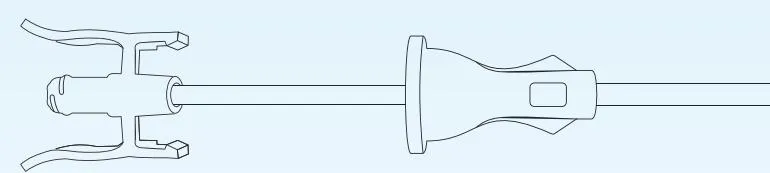
| Size (Fr) | Available Models / Codes |
|---|---|
| 10Fr | H04-1013, H04-1020, H04-1028, H04-1035, H04-1045, H04-1055 |
| 12Fr | H04-1213, H04-1220, H04-1228, H04-1235, H04-1245, H04-1255 |
| 14Fr | H04-1413, H04-1420, H04-1428, H04-1435, H04-1445, H04-1455 |



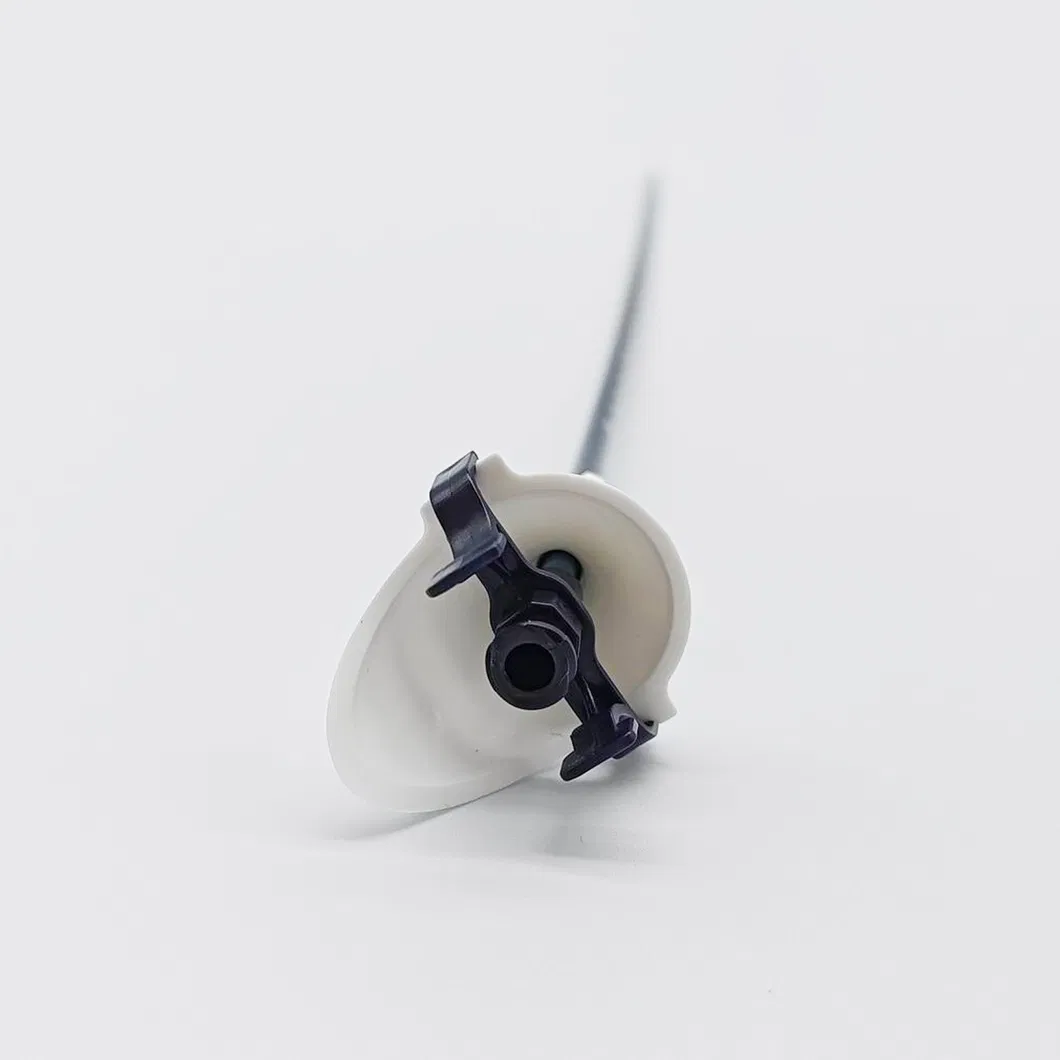


Used during urological endoscopy to build a channel via guide-wire intervention, facilitating access to the renal pelvis for endoscopes or other surgical instruments. Designed for short-term, single use.
Cautions