
1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| OEM: | Accept OEM |
| Tolerance: | +/-0.02mm |
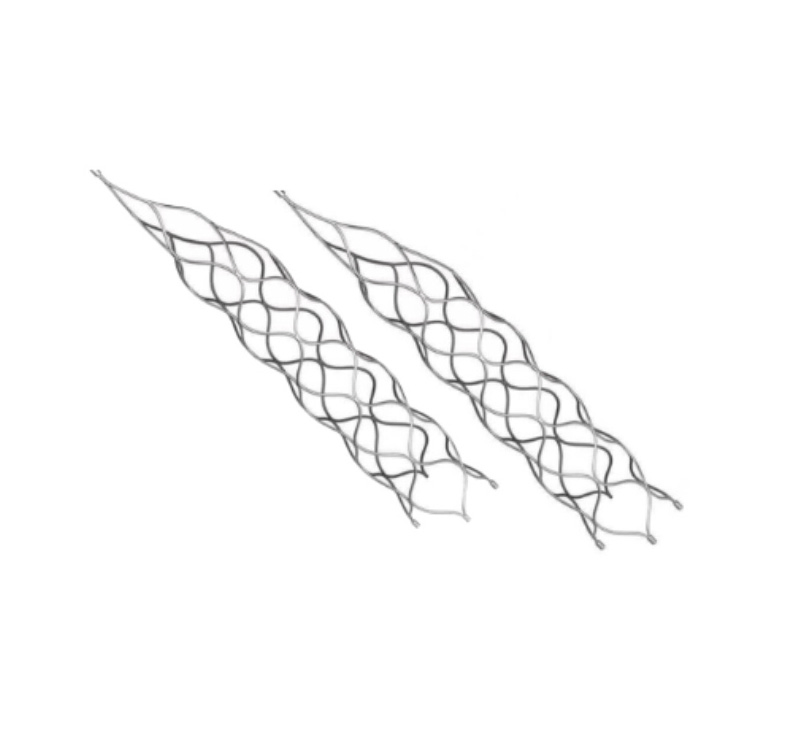
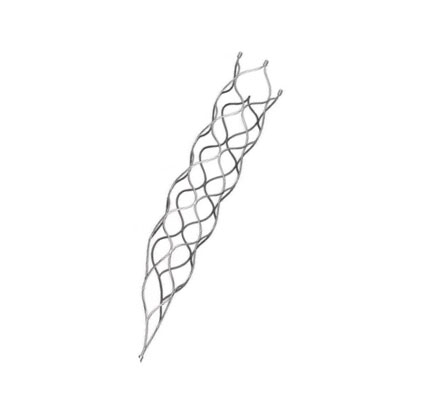
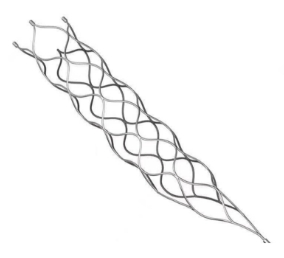
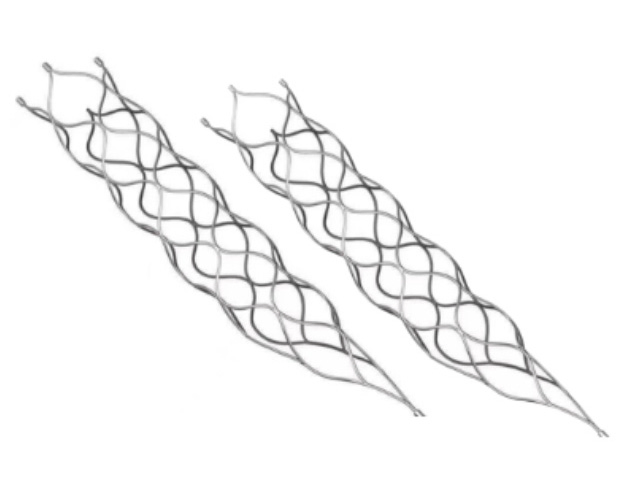
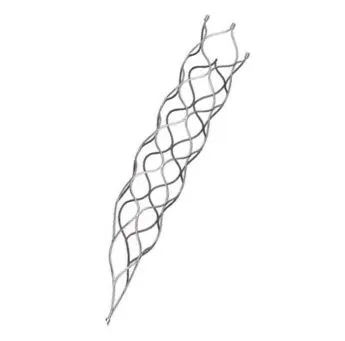
This medical device is specifically designed for the treatment of vascular diseases. The primary material utilized is high-grade nickel-titanium (Nitinol) tubing, ensuring excellent biocompatibility and performance.
Application Scope: This product is optimized for professional surgical applications.
Established in 2019, our company is a comprehensive enterprise integrating medical device research, development, production, and sales services. We operate under the ISO13485:2016 quality system certification.
Our facilities span over 5,000 square meters with a dedicated workforce of more than 100 professionals across specialized processing workshops.









