
1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| OEM: | Accept OEM |
| Tolerance: | +/-0.02mm |
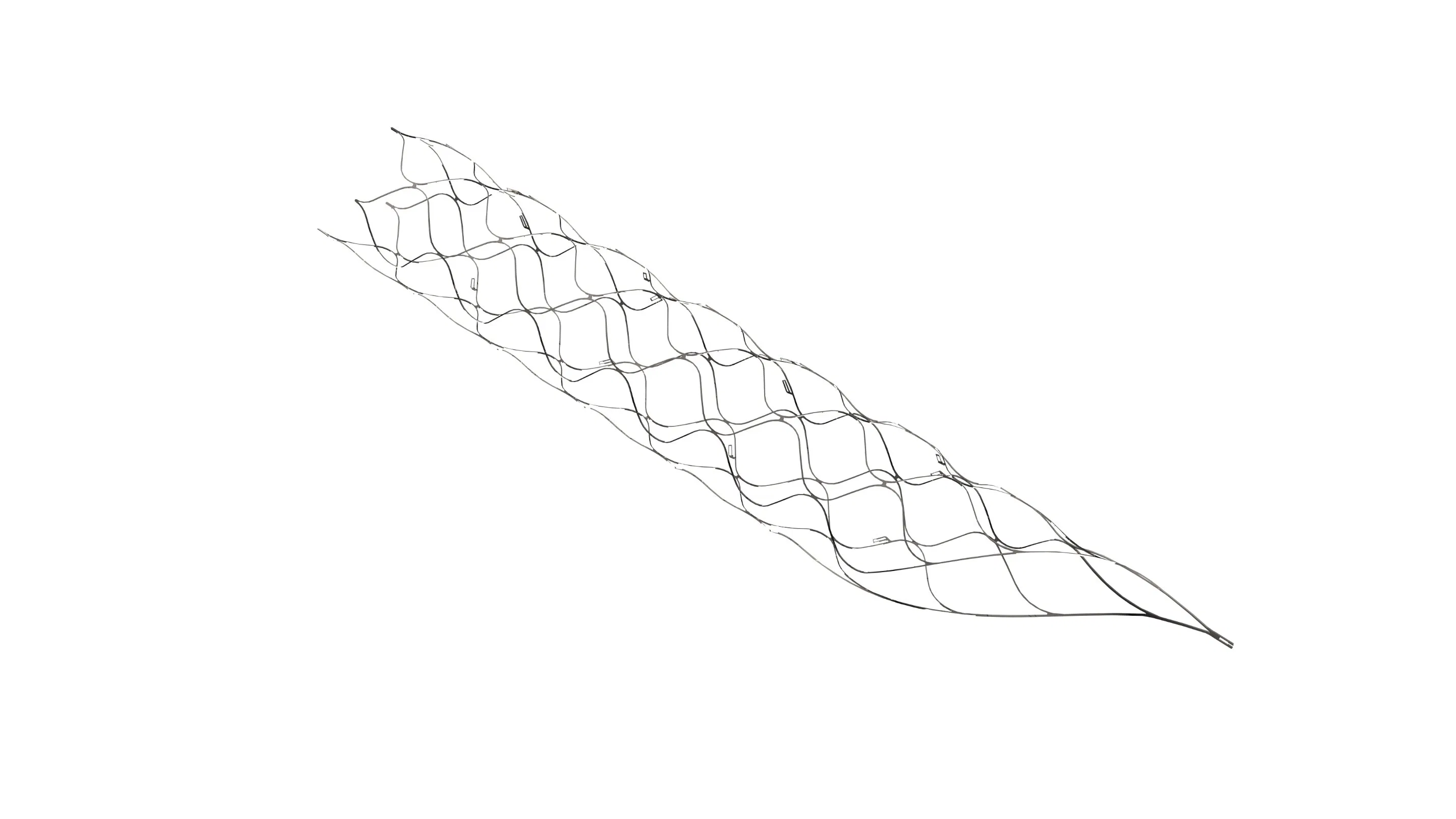
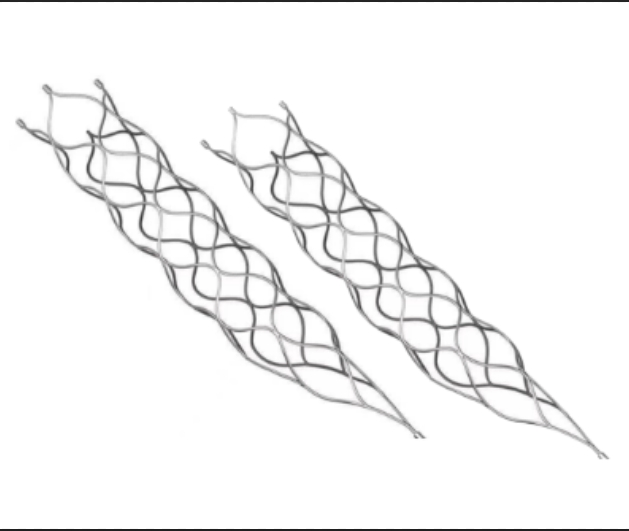
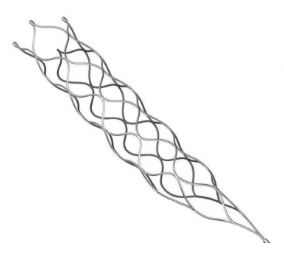
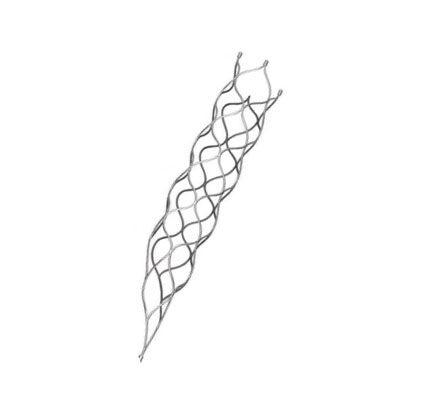
This medical device is specifically engineered for the treatment of vascular diseases. The primary construction utilizes high-grade nickel-titanium (Nitinol) tubing, ensuring excellent biocompatibility and performance.
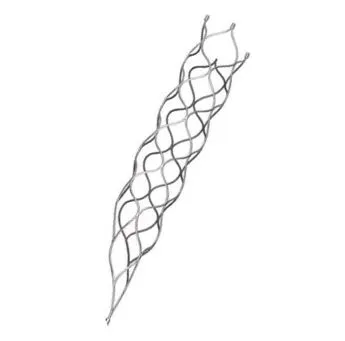
Our facility integrates research, development, and production with over 5,000 square meters of specialized workshops. We maintain a quality system certified to high international standards to ensure medical-grade precision.









