
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| Function: | Improve Blood Circulation |
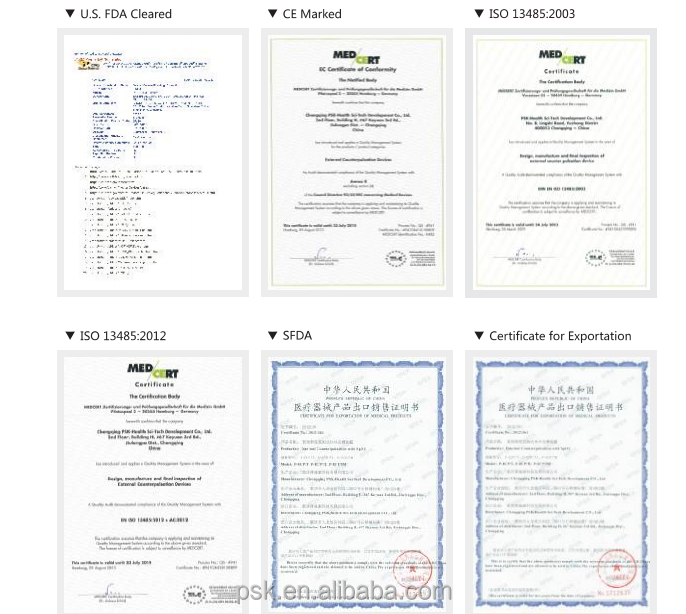
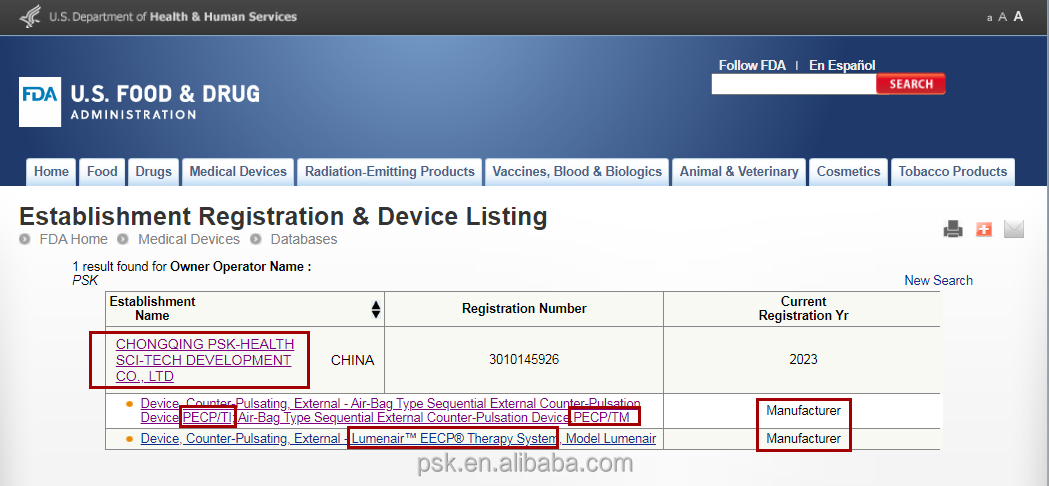
| Certification: | CE, FDA, ISO13485 |
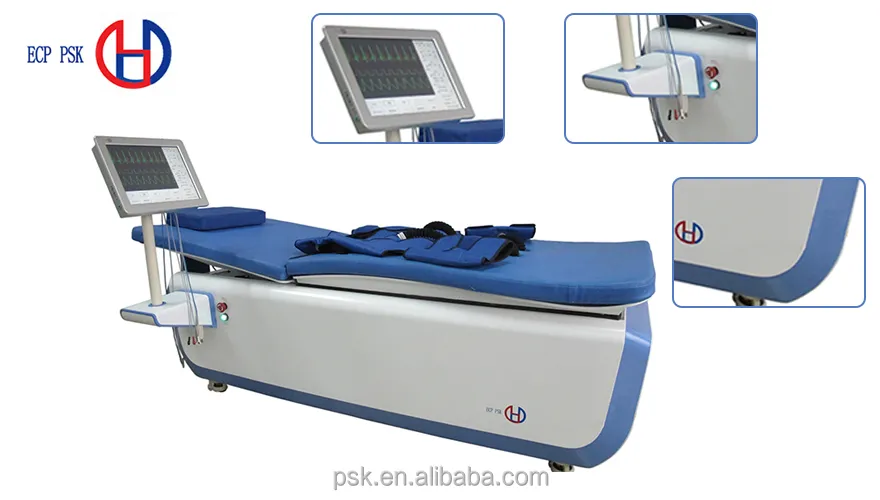
This high-tech enterprise is dedicated to the research, development, manufacturing, and distribution of EECP and other cardiac medical devices. As a leading manufacturer of EECP devices, the company serves as the drafting unit for National EECP Production Standards. With a global presence, professional medical adviser teams provide prompt and professional service worldwide. The EECP devices have been exported to thirty-five countries, maintaining a dominant market share both domestically and internationally.

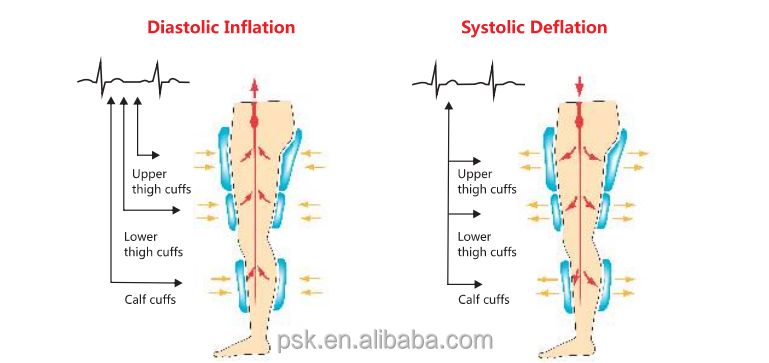
EECP is a non-invasive therapy for assisting circulation. Its mechanism uses the R-wave of a human ECG as a trigger signal to sequentially inflate and deflate during diastole and systole. During diastole, three sets of cuffs wrapped around limbs and buttocks inflate, forcing arterial blood toward the aorta to increase blood perfusion in vital organs like the heart, brain, and kidneys. During systole, the cuffs deflate to decrease the heart's workload and facilitate blood ejection. This process repeats with the cardiac cycle to achieve therapeutic goals.
| Pressure Setting | 1kPa-50kPa |
| Setting Step | 1kPa |
| Treatment Time Setting | 1-60min |
| Solenoid Valve Response Time | ≤40ms |
| Load-bearing Capacity | 150kg |
| Adjustable Backrest Angle | 0-40° |
| Safety Protection | Heart Rate, Premature Beat, Emergency Stop, and Operating Pressure Protection available. |
| Counter-pulsation Ratio | 1:1 or 1:2 |


The device is secured in a fumigated wooden case (75kg) to ensure safe, damage-free delivery.