1 / 5
 SJ Medical
SJ Medical










| Still deciding? Get samples of $ ! US$ 480/kg Request Sample |
| Product Name | Bivalirudin Powder |
| Certification | GMP, HSE, ISO 9001, USP, BP |
| CAS No. | 128270-60-0 |
| Molecular Formula | C98H138N24O33 |
| Purity | >99% (HPLC) |
| Appearance | White Powder |
| Grade | Pharmaceutical Grade |
| Shelf Life | 2 Years |
| State | Powder |

Bivalirudin is a synthetic 20 amino acid peptide direct thrombin inhibitor presented as a white to off-white lyophilized powder. It is highly soluble in water and in physiological saline solutions. The peptide sequence is H-D-Phe-Pro-Arg-Pro-Gly-Gly-Gly-Gly-Asn-Gly-Asp-Phe-Glu-Glu-Ile-Pro-Glu-Glu-Tyr-Leu-OH with a molecular weight of approximately 2180 Daltons.
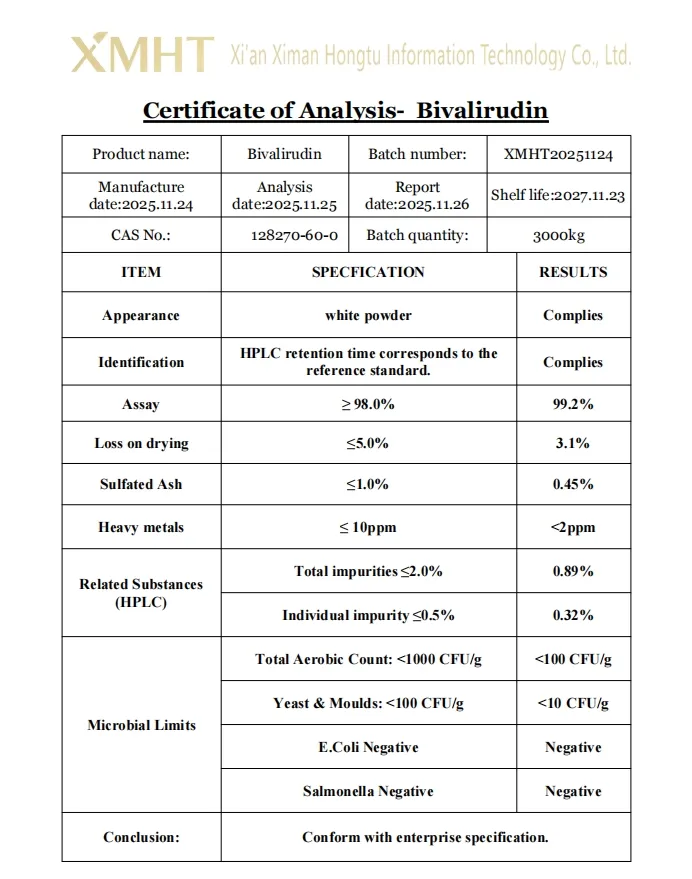
Identity is confirmed by methods such as HPLC and peptide mapping using mass spectrometry. Purity is determined by reversed-phase HPLC, typically not less than 98.0 percent. Water content is strictly controlled via Karl Fischer titration (usually not more than 8.0 percent), and bacterial endotoxins are limited to ensure pharmaceutical safety.



| Product Name | Bivalirudin |
| Appearance | White Powder |
| CAS | 128270-60-0 |
| MF | C98H138N24O33 |
| MW | 2180.317 |