 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
| Type: | Coronary Stents |
| Material: | Fully Degradable Poly(L-Lactide) |
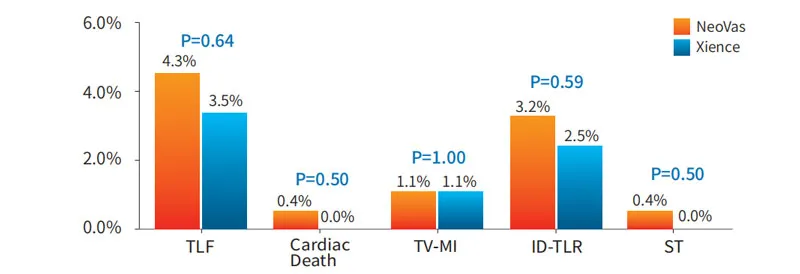
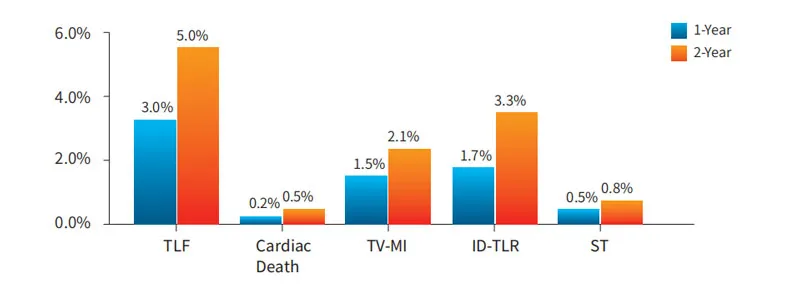
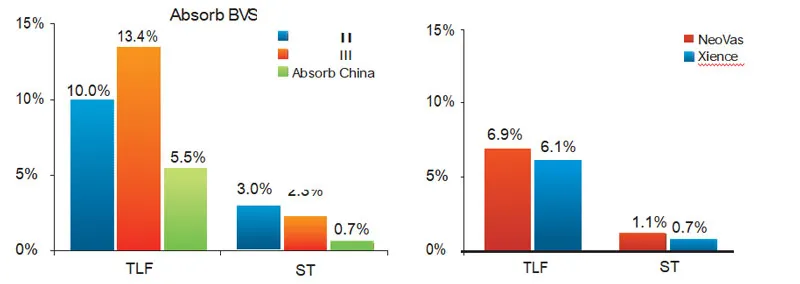
The study enrolled 560 subjects in 33 centers. Test group (NeoVas): 278 cases; Control group (Xience stent): 282 cases. Primary endpoint: late lumen loss in one-year follow-up.

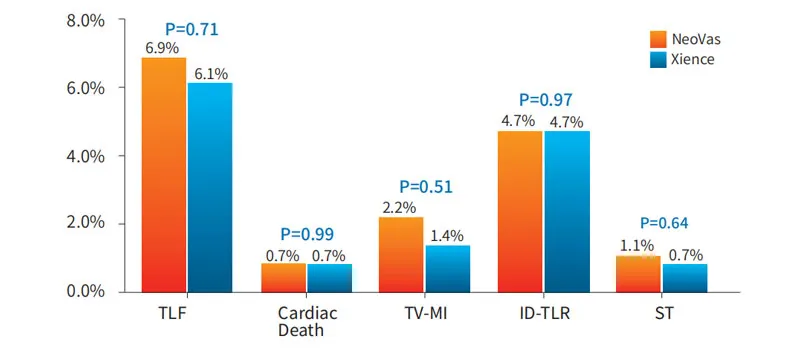
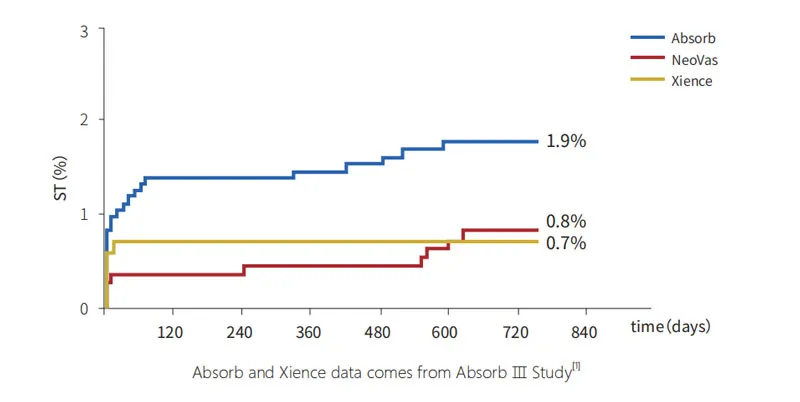
Enrolled 1103 subjects across 45 centers. Results demonstrate that NeoVas bioresorbable stent has a favorable safety profile compared to the first-generation BVS, with significantly decreased clinical event rates.
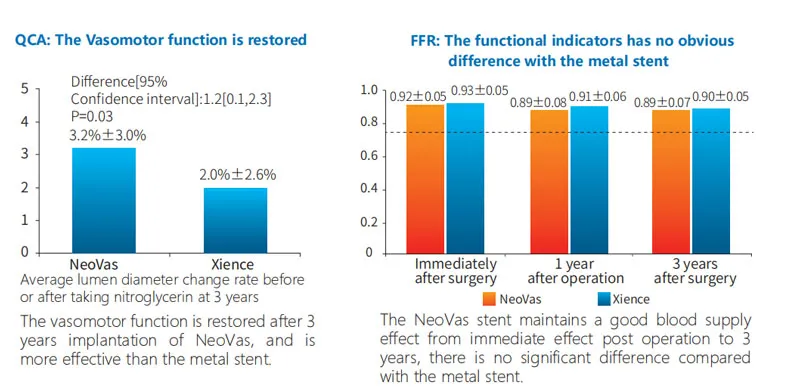
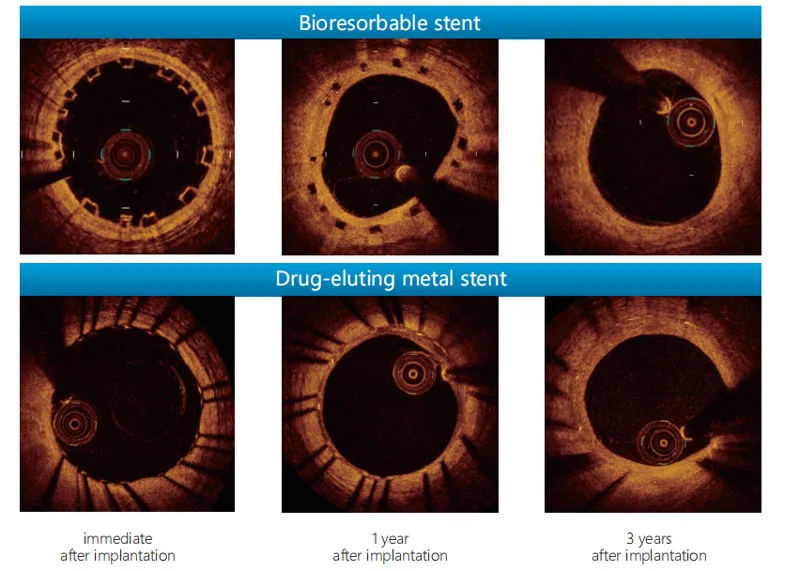
OCT data at 3 years shows vessel vasomotor function restoration. Most of the stent was successfully degraded and absorbed after 3 years, maintaining a favorable level of blood supply.
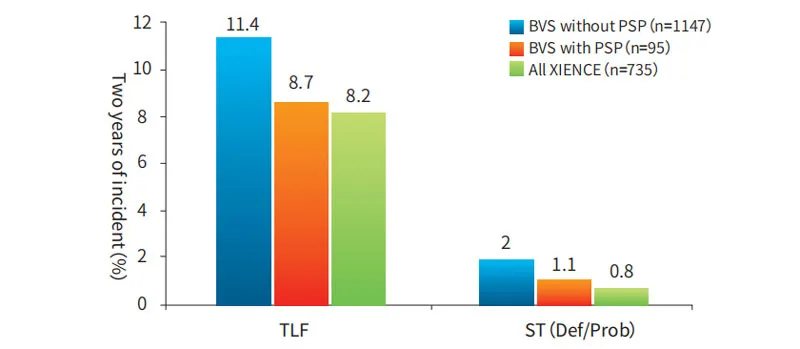
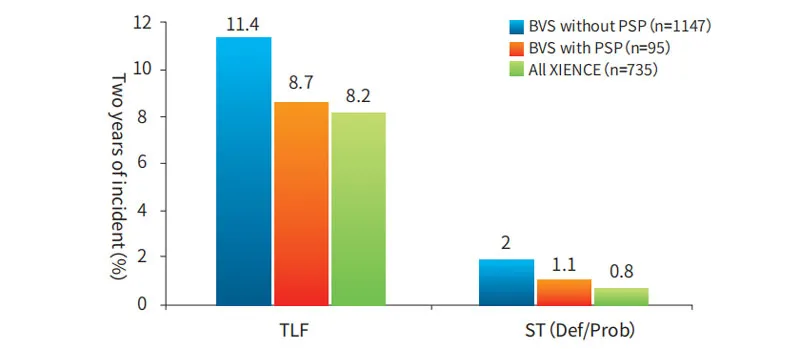
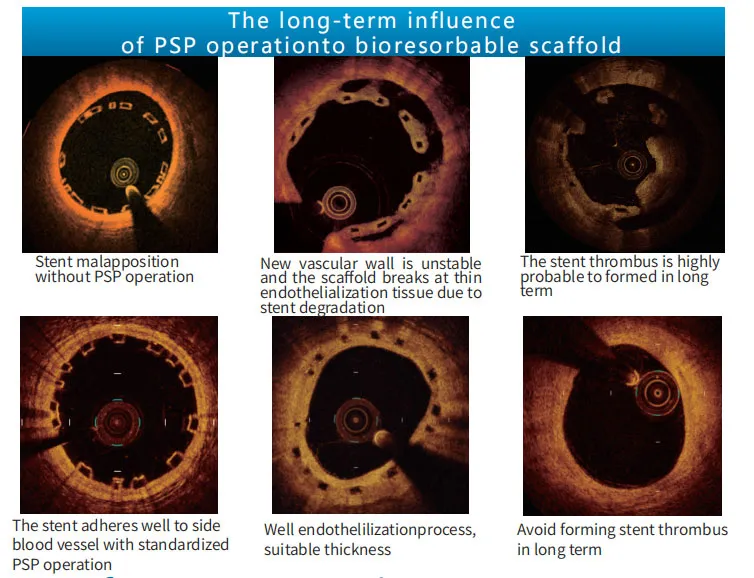
Standardized PSP (Pre-dilatation, Sizing, Post-dilatation) operation ensures proper stent apposition and excludes vascular dissection. Failure to perform PSP can increase the risk of stent thrombus and TLF (Target Lesion Failure).
Operation Flowchart:

| Stent Diameter (mm) | Stent Length (mm) | ||||
|---|---|---|---|---|---|
| 12 | 15 | 18 | 21 | 24 | |
| 2.75 | LPS2712 | LPS2715 | LPS2718 | LPS2721 | LPS2724 |
| 3.0 | LPS3012 | LPS3015 | LPS3018 | LPS3021 | LPS3024 |
| 3.5 | LPS3512 | LPS3515 | LPS3518 | LPS3521 | LPS3524 |
| Balloon Diameter (mm) | Pressure kPa (atm) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| - | 2 (203) | 4 (405) | 6 (608) | 7 (709) | 8 (810) | 10 (1013) | 12 (1216) | 14 (1418) | 16 (1621) | 18 (1824) | 20 (2027) |
| 2.75 | 2.60 | 2.66 | 2.71 | 2.73 | 2.75 | 2.82 | 2.88 | 2.94 | 3.00 | 3.06 | 3.12 |
| 3.00 | 2.84 | 2.89 | 2.95 | 2.97 | 3.00 | 3.06 | 3.12 | 3.18 | 3.25 | 3.30 | 3.36 |
| 3.50 | 3.37 | 3.42 | 3.47 | 3.50 | 3.54 | 3.61 | 3.68 | 3.75 | 3.81 | 3.88 | 3.95 |