1 / 5
 SJ Medical
SJ Medical










| Customization: | Available |
|---|---|
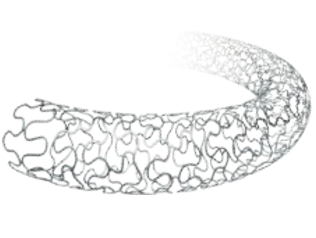
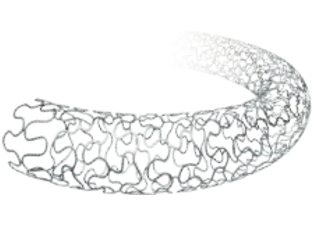
| Type: | Coronary Stents |
| Material: | 316L Stainless Steel |
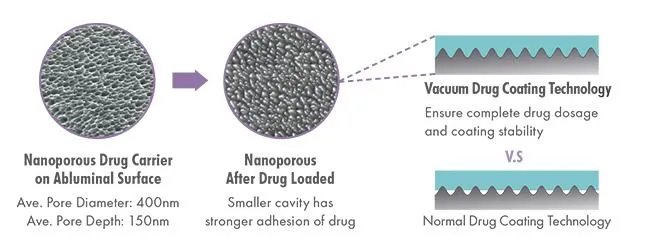

Safety & Efficacy Profile:
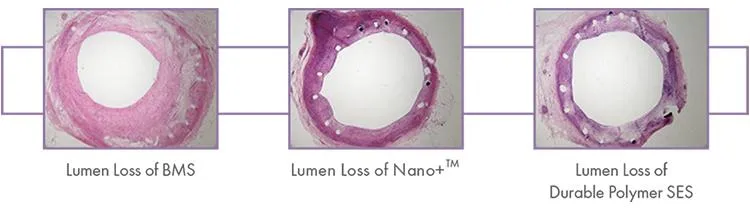
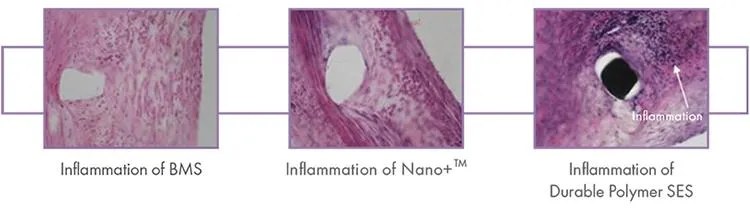
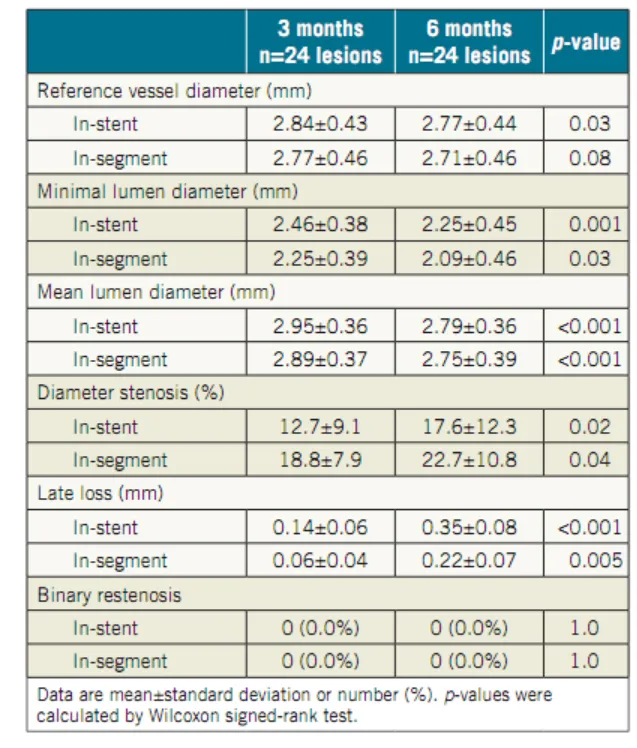
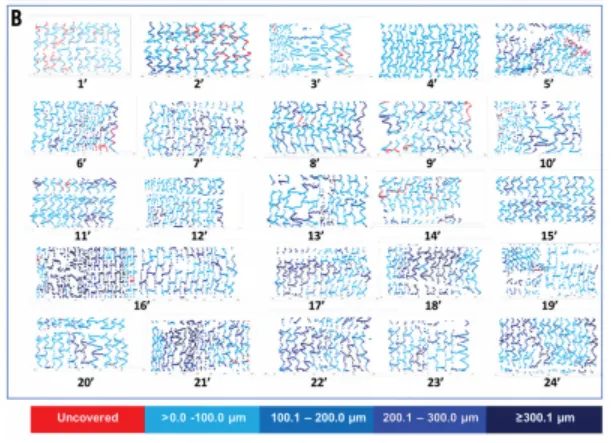
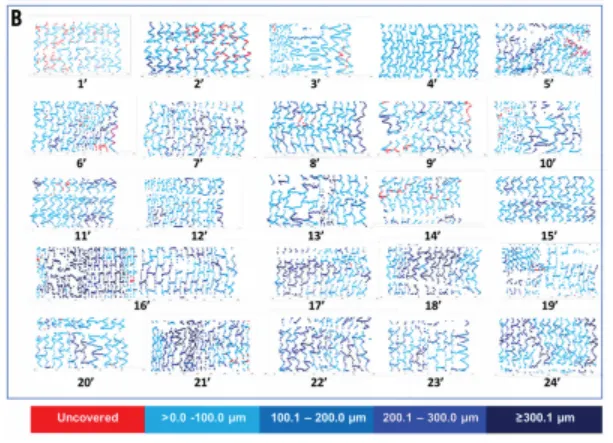
Nano+™ OCT Study: An OCT Study in Europe Shows Nano+™ A Great Combination of DES Efficacy and BMS Safety. Serial OCT showed almost complete vascular healing at six months, even when coverage was insufficient at three months.
| Parameter | Specification |
|---|---|
| Stent Material | 316L Stainless Steel |
| Strut Design | Nanoporous drug carrier on abluminal surface |
| Metal to Artery Ratio | 15% |
| Recoil | < 2% |
| Shortening | < 1% |
| Crossing Profile | 0.035″ (0.90mm) |
| Diameter (mm) | 12mm | 15mm | 18mm | 21mm | 24mm | 29mm | 33mm | 36mm |
|---|---|---|---|---|---|---|---|---|
| 2.5 | LPRPS2512 | LPRPS2515 | LPRPS2518 | LPRPS2521 | LPRPS2524 | LPRPS2529 | LPRPS2533 | - |
| 2.75 | LPRPS2712 | LPRPS2715 | LPRPS2718 | LPRPS2721 | LPRPS2724 | LPRPS2729 | LPRPS2733 | LPRPS2736 |
| 3.0 | LPRPS3012 | LPRPS3015 | LPRPS3018 | LPRPS3021 | LPRPS3024 | LPRPS3029 | LPRPS3033 | LPRPS3036 |
| 3.5 | LPRPS3512 | LPRPS3515 | LPRPS3518 | LPRPS3521 | LPRPS3524 | LPRPS3529 | LPRPS3533 | LPRPS3536 |
| 4.0 | LPRPS4012 | LPRPS4015 | LPRPS4018 | LPRPS4021 | LPRPS4024 | LPRPS4029 | LPRPS4033 | LPRPS4036 |