SJ Medical
SJ Medical










| Still deciding? Get samples of $ ! US$ 50/g Request Sample |

Tirofiban hydrochloride (CAS number: 150915-40-5) is a white or off-white powder that belongs to a class of highly effective antiplatelet drugs. As a glycoprotein IIb/IIIa receptor antagonist, it effectively prevents thrombosis by specifically blocking the final pathway of platelet aggregation.

This product is primarily used for the clinical treatment of acute coronary syndrome, such as unstable angina and percutaneous coronary intervention (PCI). It serves as a critical injectable raw material in the field of cardiovascular emergency treatment.
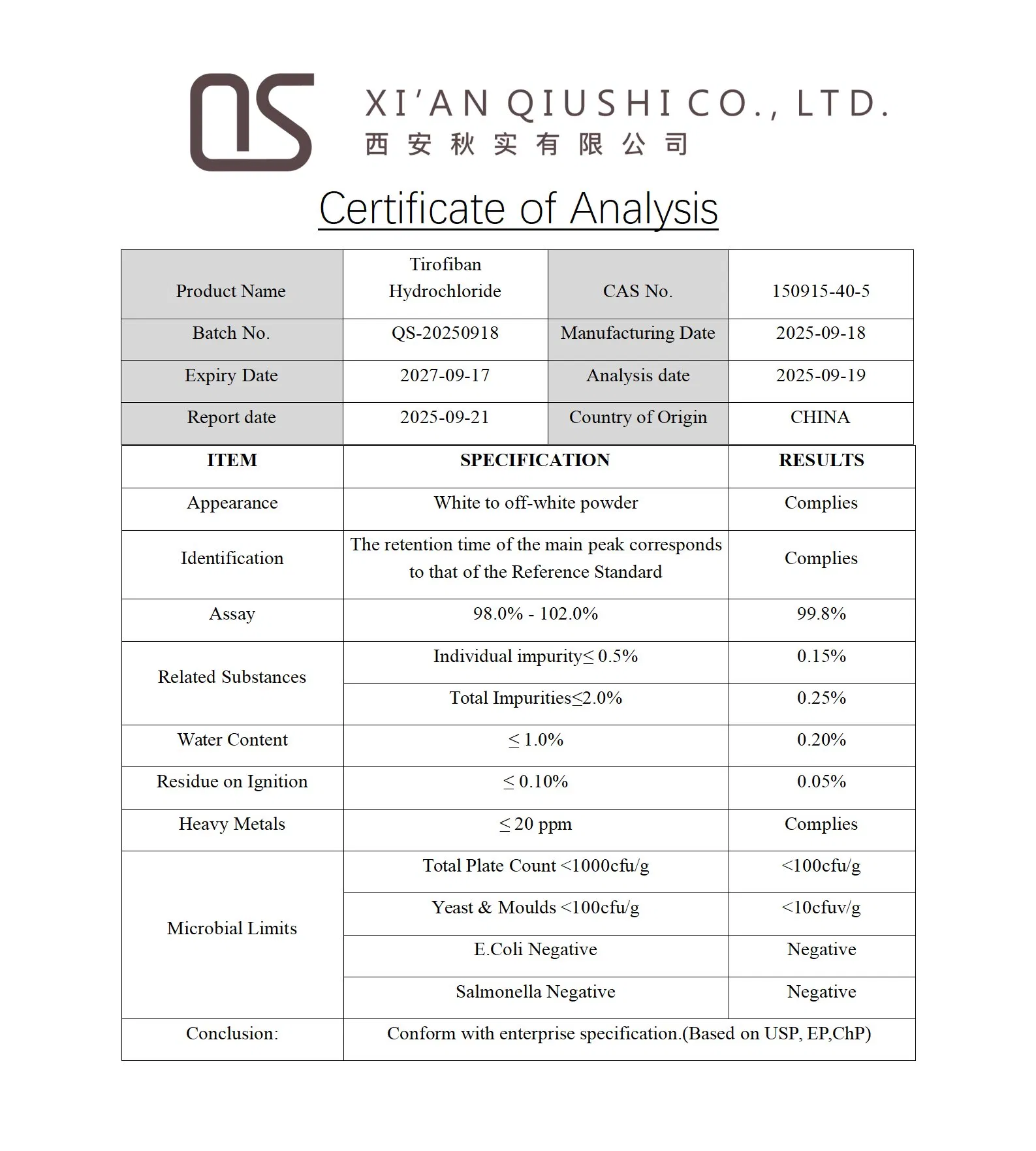
| Product Name | Tirofiban hydrochloride |
| Appearance | White to off-white powder |
| Melting Point | 135-137°C |
| Boiling Point | 611.7 °C at 760 mmHg |
| Assay | 99% min |
| Test Method | HPLC |
The mechanism of action lies in its ability as a highly selective and reversible non-peptide small molecule to competitively antagonize glycoprotein IIb/IIIa receptors on the surface of platelet membranes. This receptor is the ultimate common pathway for platelet aggregation.
By occupying the receptor position, it effectively blocks the bridging effect of adhesion proteins like fibrinogen, thereby inhibiting platelet cross-linking. This rapid action prevents blood clot expansion, maintains vascular patency, and improves patient prognosis in acute cardiovascular events.