SJ Medical
SJ Medical










| Still deciding? Get samples of $ ! US$ 50/g Request Sample |

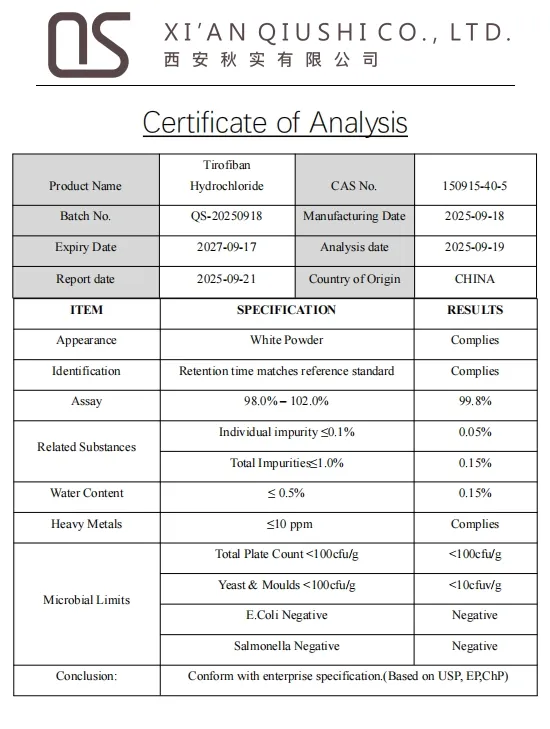
| Certification | GMP, HSE, ISO 9001, USP, BP | Suitable for | Elderly, Children, Adult |
| State | Powder | Purity | >99% |
| Product Name | Tirofiban Hydrochloride | CAS | 150915-40-5 |
| Molecular Formula | C22H39ClN2O6S | Molecular Weight | 495.07286 |
| Application | Pharmaceutical | Appearance | White Powder |
| Density | 1.28 | Melting Point | 135 |
| Assay | 99% | Capacity | 1000kg/Month |


Tirofiban Hydrochloride is a non-peptide platelet glycoprotein Ⅱb/Ⅲa receptor antagonist. Its core mechanism of action is to specifically and competitively bind to glycoprotein Ⅱb/Ⅲa receptors on the platelet surface, blocking the binding of ligands such as fibrinogen and von Willebrand factor to this receptor — the glycoprotein Ⅱb/Ⅲa receptor is the key "bridge" for platelet aggregation; once blocked, it can strongly inhibit the activation and aggregation of platelets, thereby preventing thrombosis and improving vascular blood perfusion.

Tirofiban Hydrochloride is primarily used in the management of acute coronary syndromes (ACS) and to prevent thrombotic complications during percutaneous coronary intervention (PCI). It targets the final common pathway of platelet aggregation.
When platelets are activated during vascular injury, the GP Ⅱb/Ⅲa receptor binds to fibrinogen or von Willebrand factor (vWF), forming thrombus. Tirofiban Hydrochloride competitively binds to these receptors with high affinity, blocking the interaction and completely preventing platelet cross-linking. This action maintains coronary artery patency and protects myocardial tissue from ischemic damage.