 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
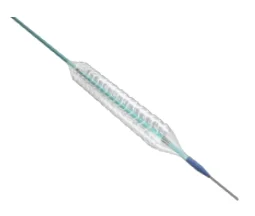 Sirolimus-Eluting Bioresorbable Coronary Stent Implant
Sirolimus-Eluting Bioresorbable Coronary Stent Implant
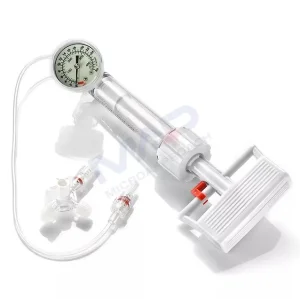 Balloon Expansion Pressure Pump Balloon Inflation Device Percutaneous Transluminal Coronary Angioplasty Ptca Surgery
Balloon Expansion Pressure Pump Balloon Inflation Device Percutaneous Transluminal Coronary Angioplasty Ptca Surgery
 Dedicated Vascular Balloon Catheter Angioplasty Stent Components Chocolate Balloon
Dedicated Vascular Balloon Catheter Angioplasty Stent Components Chocolate Balloon
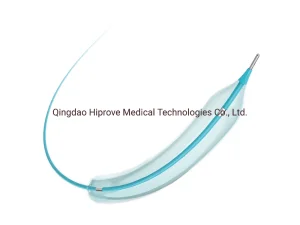 Interventional Materials Balloon Dilatation Catheters Angioplasty Stent Ptca balloon Catheter
Interventional Materials Balloon Dilatation Catheters Angioplasty Stent Ptca balloon Catheter
 High Pressure/Nc Balloon Dilatation Angioplasty Stent Medical Cardiology Interventional Surgical Coronary Heart Disease Ptca Balloon Catheter
High Pressure/Nc Balloon Dilatation Angioplasty Stent Medical Cardiology Interventional Surgical Coronary Heart Disease Ptca Balloon Catheter
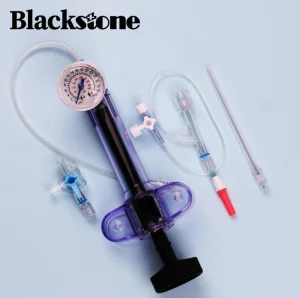 Disposable Supply Inflator Balloon Catheter Renal Artery Angioplasty Ptca Balloon Inflation Device
Disposable Supply Inflator Balloon Catheter Renal Artery Angioplasty Ptca Balloon Inflation Device
 Advanced Percutaneous Transluminal Angioplasty Balloon Catheter for Optimal Vascular Access
Advanced Percutaneous Transluminal Angioplasty Balloon Catheter for Optimal Vascular Access
 Disposable Pigtail Catheter Transplant Kidney Stent EOS Sterilization for Urology Use
Disposable Pigtail Catheter Transplant Kidney Stent EOS Sterilization for Urology Use
The global healthcare landscape is witnessing a transformative shift toward minimally invasive surgeries, with the Balloon Expandable Stent market at the forefront of this revolution. As cardiovascular diseases (CVDs) remain the leading cause of mortality worldwide, the demand for high-precision interventional devices has surged. From the bustling hospitals of Europe to the rapidly developing healthcare systems in Asia and the Middle East, clinicians are increasingly relying on balloon-expandable technologies for their predictable radial strength and superior deliverability.
Today, the industry is not just about expanding a vessel; it’s about biocompatibility, drug-elution precision, and long-term clinical efficacy. Markets in the USA and European Union are driving innovation in thin-strut designs and bioresorbable materials, while emerging markets in India, Indonesia, and Brazil are focusing on accessibility and reliable performance. As a premier Balloon Expandable Stent Factory, we understand that "one size does not fit all." The global commercial trend is shifting toward specialized stents for peripheral, renal, and biliary applications, requiring manufacturers to possess deep technical expertise and agile production capabilities.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes. This ensures that every balloon expandable stent and catheter produced meets the stringent safety requirements of the medical industry.
Our facility is equipped with state-of-the-art precision balloon forming equipment, laser welding, and marking technologies. We maintain strict quality control through comprehensive laboratory testing facilities, ensuring high levels of manufacturing accuracy and product consistency. Under the leadership of senior engineers with decades of experience, we continuously improve our processes to support global procurement needs for high-quality interventional devices.
Utilizing high-precision laser cutting for Ni-Ti and Cobalt-Chromium stents, ensuring smooth surfaces and optimal radial force.
Our technical team focuses on polymer science and drug-eluting coatings to enhance clinical outcomes and reduce restenosis rates.
Strict adherence to CE, ISO 13485, and CFDA standards, providing full traceability and regulatory confidence for international exporters.
The next decade of vascular intervention will be defined by Smart Stents and Bioresorbable Scaffolds. There is a growing demand for "leave-nothing-behind" technologies. Furthermore, the integration of AI in diagnostic imaging is requiring stents to be more radiopaque and visible under advanced fluoroscopy. Our R&D department is currently exploring these frontiers, ensuring that our OEM/ODM partners stay ahead of the curve.
As a leading China Balloon Expandable Stent Factory, SJ Medical offers a unique blend of cost-efficiency, rapid prototyping, and massive scalability. The Chinese medical device supply chain has matured into a global hub, allowing us to source premium raw materials and combine them with lower manufacturing overheads. This translates to higher value for hospitals and distributors without compromising on the rigorous quality standards expected in the EU or North America.
Whether it is the "Make in India" initiative or the localization requirements in Southeast Asia, SJ Medical provides flexible OEM/ODM services to help brands enter local markets. We offer customized labeling, specific catheter lengths, and proprietary balloon diameters to meet local clinical preferences in countries like Italy, Spain, Indonesia, and Turkey.

Our products are widely applied in minimally invasive procedures involving both coronary and peripheral vascular systems. They play an important role in the diagnosis and treatment of vascular diseases, helping to improve procedural efficiency while enhancing patient safety and recovery.
SJ Medical Global Footprint: We have established long-term partnerships with clients across Europe, Asia, the Middle East, and Africa. Our products are currently distributed in countries such as Finland, Italy, Spain, France, the Netherlands, India, Indonesia, Turkey, and Egypt. We provide comprehensive support, from pre-sales consultation to efficient after-sales technical assistance.
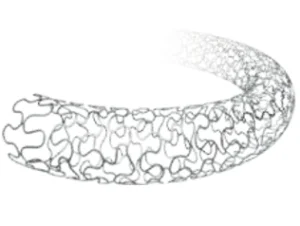 Polymer-Free Sirolimus-Eluting Coronary Stent System
Polymer-Free Sirolimus-Eluting Coronary Stent System
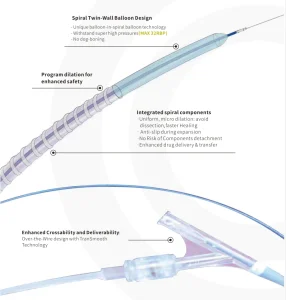 Durable Pta Spiral Balloon Catheter for Enhanced Vascular Procedures
Durable Pta Spiral Balloon Catheter for Enhanced Vascular Procedures
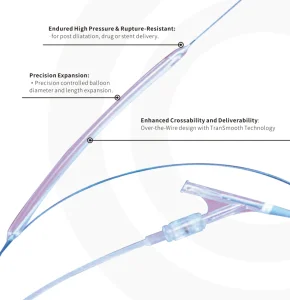 Precision Engineered Pta Balloon Dilatation Catheter for Cardiac Procedures
Precision Engineered Pta Balloon Dilatation Catheter for Cardiac Procedures
 OEM Custom Processing Medical Laser Cutting Ni-Ti Thrombectomy Stent
OEM Custom Processing Medical Laser Cutting Ni-Ti Thrombectomy Stent
 Premium High Pressure Balloon Dilatation Catheter for Enhanced Vascular Procedures
Premium High Pressure Balloon Dilatation Catheter for Enhanced Vascular Procedures
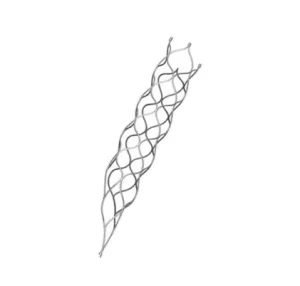 High-Precision Customized Laser-Cut Thrombectomy Stent
High-Precision Customized Laser-Cut Thrombectomy Stent
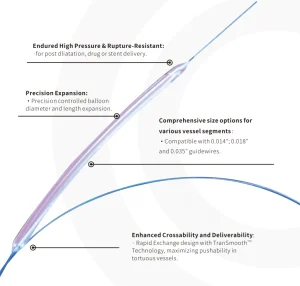 Reliable Pta Balloon Dilatation Catheter for Enhanced Patient Outcomes
Reliable Pta Balloon Dilatation Catheter for Enhanced Patient Outcomes
 Precision Vascular Interventional Surgery Medical Device Thrombectomy Stent
Precision Vascular Interventional Surgery Medical Device Thrombectomy Stent
The manufacturing of a Balloon Expandable Stent is a feat of micromechanical engineering. At SJ Medical, we utilize high-grade materials such as 316L Stainless Steel and Cobalt-Chromium (CoCr) alloys. CoCr stents allow for thinner struts without sacrificing radial strength, which is critical for reducing vessel wall injury and improving healing. Our OEM services allow clients to specify strut thickness and stent geometry to optimize for specific clinical indications, such as calcified lesions or bifurcations.
Furthermore, the PTCA Balloon Catheter technology that delivers these stents must be equally precise. Our NC (Non-Compliant) and semi-compliant balloons are engineered to provide uniform expansion, preventing "dog-boning" and ensuring the stent is perfectly apposed to the vessel wall. This level of detail is why SJ Medical is a trusted name for Global Exporters looking for reliable interventional supplies.
In terms of Global Procurement Demand, we have seen a massive rise in "bulk procurement" from government health tenders in Asia and South America. These regions require high-quality products that meet CE and ISO certifications but at a price point that allows for widespread public health use. SJ Medical’s ability to scale production while maintaining localized "hundred-class" cleanroom standards makes us the ideal partner for these large-scale interventional projects.
Our commitment to E-E-A-T principles means we don't just sell products; we share expertise. We provide white papers on balloon compliance, clinical data on our Sirolimus-eluting coatings, and technical training for hospital staff. This transparency builds trustworthiness and establishes SJ Medical as an authoritative leader in the vascular intervention field.
Looking ahead, we are expanding our peripheral intervention line, focusing on PTA balloon catheters and stents designed for the unique challenges of the iliac, femoral, and popliteal arteries. By understanding the mechanical stresses of the peripheral system—including torsion, compression, and flexion—we are designing the next generation of durable interventional tools.