 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
 Hemodialysis Machine in Nephrology Dept
Hemodialysis Machine in Nephrology Dept
 Crutches Hospital Furniture Medical Underarm Crutches for Patient
Crutches Hospital Furniture Medical Underarm Crutches for Patient
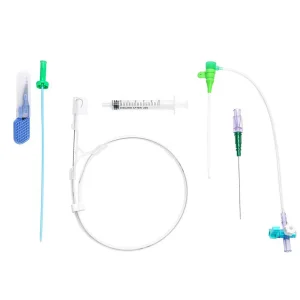 Premium Vascular Access Catheter Sheath
Premium Vascular Access Catheter Sheath
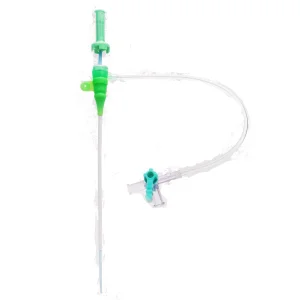 Premium Hospital-Grade Medical Catheter Sheath
Premium Hospital-Grade Medical Catheter Sheath
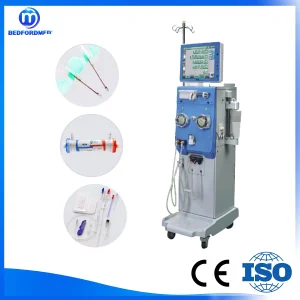 Medical Hemodialysis Machine with CE
Medical Hemodialysis Machine with CE
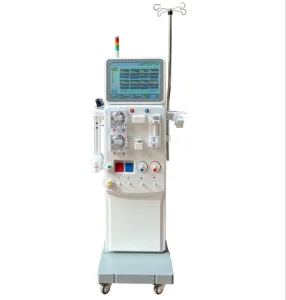 Hemodialysis Machines for Kidney Care
Hemodialysis Machines for Kidney Care
 Aluminum Adjustable Walking Stick
Aluminum Adjustable Walking Stick
 Health Care Adjustable Walking Aids
Health Care Adjustable Walking Aids
The global healthcare landscape has undergone a paradigm shift in its approach to infection control. As a premier China Hospital Disinfection Robot Factory, we have witnessed the exponential growth of the "No-Touch Disinfection" (NTD) market. Currently, the global market for autonomous disinfection robots is projected to grow at a CAGR of over 25%, driven by the urgent need to mitigate Healthcare-Associated Infections (HAIs).
In modern medical facilities, the reliance on manual cleaning is no longer sufficient. Human error, inconsistent application of disinfectants, and the emergence of multi-drug resistant organisms (MDROs) have made Autonomous Mobile Robots (AMRs) a necessity rather than a luxury. From UVC radiation to dry mist hydrogen peroxide (H2O2), these robots provide a level of consistency and efficacy that manual labor cannot match.
Modern robots utilize Simultaneous Localization and Mapping (SLAM) and LIDAR sensors to navigate complex hospital corridors without human intervention, avoiding dynamic obstacles seamlessly.
The trend is moving toward hybrid systems that combine 254nm UVC light with ultrasonic dry mist spray, ensuring both air and surface pathogens are eradicated.
Data-driven disinfection reports are now standard. Our robots generate digital logs of every cycle, providing hospitals with the "Proof of Disinfection" required for audit compliance.
Why do international healthcare groups look for a China Hospital Disinfection Robot Supplier? The answer lies in the unique combination of high-tech integration and supply chain efficiency. Procurement departments in Europe, the Middle East, and Southeast Asia are prioritizing "Total Cost of Ownership" (TCO).
Key procurement requirements we fulfill include:
As a leading manufacturer, SJ Medical addresses these needs by offering OEM/ODM services that allow regional distributors to localize the software and branding while maintaining world-class hardware standards.
In high-risk zones, our robots provide rapid turnaround disinfection between patients, reducing the risk of cross-contamination significantly.
UVC robots ensure shadowless sterilization of surgical equipment and floors, reaching areas often missed by manual wiping.
Continuous air purification modes allow robots to operate safely around people in lobbies, using HEPA filtration and shielded UVC.
Hangzhou SJ Medical Co., Ltd. is not just a trading company; we are a specialized manufacturer with a deep-rooted history in high-precision medical devices. Our transition into the robotics space was born from our expertise in vascular interventional medical devices and sterile environment requirements.
Our advantage as a China Disinfection Robot Factory includes:
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
The company is supported by a highly experienced technical team, many of whom have been engaged in the medical device industry for decades. Under the leadership of senior engineers, SJ Medical maintains strict quality control and continuously improves its manufacturing processes to ensure product reliability and consistency.
SJ Medical focuses on the development of vascular interventional devices designed for both coronary and peripheral procedures. Its product range integrates interventional access systems, diagnostic catheters, and therapeutic devices, forming a complete solution for minimally invasive vascular treatments. Each product is developed with an emphasis on precision, safety, and clinical performance, aiming to support physicians in achieving optimal procedural outcomes.
The company’s products are widely applied in minimally invasive procedures involving both coronary and peripheral vascular systems. They play an important role in the diagnosis and treatment of vascular diseases, helping to improve procedural efficiency while enhancing patient safety and recovery.
SJ Medical strictly adheres to international quality standards and regulatory requirements. The company has obtained CE certification, ISO 13485 quality management system certification, and CFDA approval, along with free sale certificates, demonstrating its strong commitment to product quality and compliance.
The company is equipped with advanced production and inspection systems, including precision balloon forming equipment, laser welding and marking technologies, and comprehensive laboratory testing facilities. These capabilities ensure high levels of manufacturing accuracy, product consistency, and full-process quality control.
SJ Medical has established long-term partnerships with clients across Europe, Asia, the Middle East, and Africa. Its products are currently distributed in countries such as Finland, Italy, Spain, France, the Netherlands, India, Indonesia, Turkey, and Egypt, among others. The company continues to expand its international footprint and strengthen its global presence.
SJ Medical is committed to providing comprehensive and professional services throughout the entire customer journey. From pre-sales consultation and technical support to efficient order processing and responsive after-sales service, the company ensures that every customer receives timely assistance and reliable solutions.
