 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
 Premium Intracranial Thrombosis Catheter for Safe Aspiration Procedures
Premium Intracranial Thrombosis Catheter for Safe Aspiration Procedures
 Advanced PTA Balloon Catheter for Optimal Vascular Access
Advanced PTA Balloon Catheter for Optimal Vascular Access
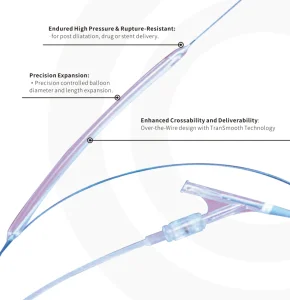 Precision Engineered PTA Balloon Dilatation Catheter
Precision Engineered PTA Balloon Dilatation Catheter
 Minimally Invasive Coronary Micro Catheter & Equipment
Minimally Invasive Coronary Micro Catheter & Equipment
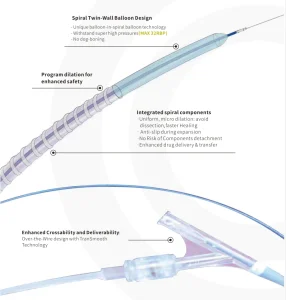 Durable PTA Spiral Balloon Catheter for Vascular Procedures
Durable PTA Spiral Balloon Catheter for Vascular Procedures
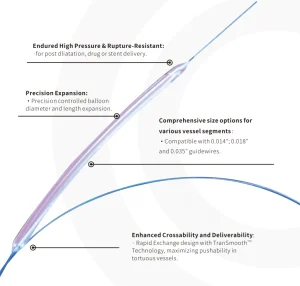 Reliable PTA Balloon Dilatation Catheter for Patient Outcomes
Reliable PTA Balloon Dilatation Catheter for Patient Outcomes
 Premium High Pressure Balloon Dilatation Catheter
Premium High Pressure Balloon Dilatation Catheter
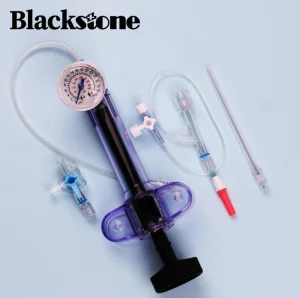 Disposable Supply Inflator Balloon Catheter PTCA Device
Disposable Supply Inflator Balloon Catheter PTCA Device
The global Intravascular Ultrasound (IVUS) market has witnessed exponential growth as clinicians shift from angiography-only approaches to imaging-guided interventions. As of 2024, the IVUS catheter industry is not just about imaging; it's about precision diagnostics that reduce stent thrombosis and optimize vessel diameter measurements.
The global market is projected to reach over $1.2 Billion by 2030, driven by an aging population and the increasing prevalence of Coronary Artery Disease (CAD).
Modern IVUS systems are incorporating Artificial Intelligence to automate plaque characterization and lumen measurements, reducing physician workload.
While the US and Japan remain leaders, the Asia-Pacific region—led by China and India—is seeing the fastest adoption of IVUS-guided PCI.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems.

SJ Medical operates a modern manufacturing facility built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes. The company is supported by a highly experienced technical team, many of whom have been engaged in the medical device industry for decades.
Precision balloon forming equipment, laser welding, and high-accuracy marking technologies ensure consistency in every catheter produced.
Comprehensive laboratory testing facilities monitor every step, from raw material inspection to final product sterilization.
Strict adherence to ISO 13485, CE certification, and CFDA approval ensures our products meet the highest global regulatory standards.
Global medical device distributors and hospital procurement departments are increasingly looking for high-quality, cost-effective alternatives to the dominant market leaders. As a Chinese factory, SJ Medical offers several distinct advantages:
SJ Medical has established long-term partnerships in countries such as Italy, Spain, France, India, Indonesia, and Turkey. Our products are widely applied in minimally invasive procedures involving both coronary and peripheral vascular systems, improving procedural efficiency while enhancing patient safety.

The industry is moving toward smaller catheter profiles (down to 3F or lower) to access more distal and tortuous peripheral vessels, enabling treatment in areas previously deemed unreachable.
Integrating IVUS with Fractional Flow Reserve (FFR) sensors allows clinicians to assess both the anatomy and the physiological significance of a lesion with a single catheter insertion.
Robotic-assisted PCI is utilizing IVUS data to provide high-precision stent placement, minimizing radiation exposure for medical staff and improving accuracy.
Manufacturers are exploring eco-friendly materials and recyclable packaging to meet the growing demand for green healthcare procurement globally.
 Vascular Balloon Catheter Angioplasty Stent - Chocolate Balloon
Vascular Balloon Catheter Angioplasty Stent - Chocolate Balloon
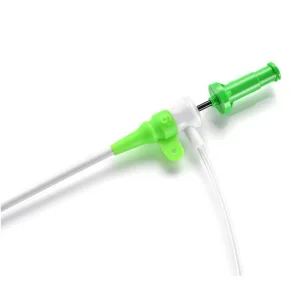 Radial Vascular Guide Wire Introduction Catheter Kit
Radial Vascular Guide Wire Introduction Catheter Kit
 Vascular Access Device Hemodialysis Catheter for Dialysis
Vascular Access Device Hemodialysis Catheter for Dialysis
 Disposable Medical Support Catheter for Peripheral Intervention
Disposable Medical Support Catheter for Peripheral Intervention
 CE Approved Radial/Femoral Coronary Guiding Catheter
CE Approved Radial/Femoral Coronary Guiding Catheter
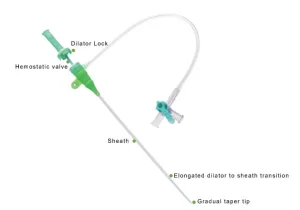 Premium Versatile Angiography Catheter Sheath - All Sizes
Premium Versatile Angiography Catheter Sheath - All Sizes
 2024 Balloon Catheter Inflation Device Manufacturer
2024 Balloon Catheter Inflation Device Manufacturer
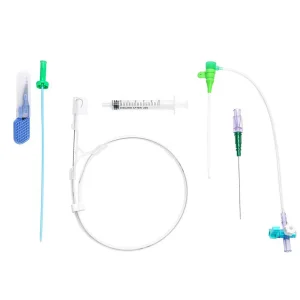 Premium Vascular Access Catheter Sheath for Hospital Use
Premium Vascular Access Catheter Sheath for Hospital Use