 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
 Medical Device Renal Access Ureteral Access Sheath
Medical Device Renal Access Ureteral Access Sheath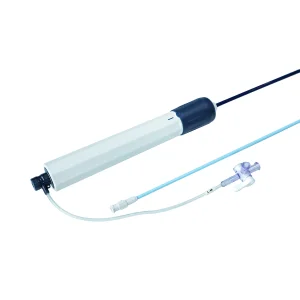 High Quality Intracardiac Access Sheath for Radiofrequency Ablation Patient
High Quality Intracardiac Access Sheath for Radiofrequency Ablation Patient Advanced Percutaneous Transluminal Angioplasty Balloon Catheter for Optimal Vascular Access
Advanced Percutaneous Transluminal Angioplasty Balloon Catheter for Optimal Vascular Access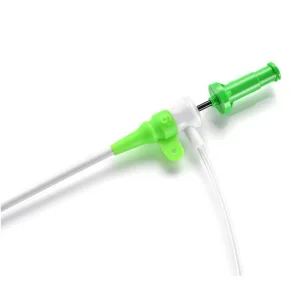 Disposable Products 4f-10f Radial Vascular Guide Wire Introduction Catheter, Ureteral Access Percutaneous Femoral Introducer Sheath Kit
Disposable Products 4f-10f Radial Vascular Guide Wire Introduction Catheter, Ureteral Access Percutaneous Femoral Introducer Sheath Kit Vascular Access Device Hemodialysis Catheter for Dialysis
Vascular Access Device Hemodialysis Catheter for Dialysis Premium Vascular Access Introducer Catheter Sheath for Hospitals
Premium Vascular Access Introducer Catheter Sheath for Hospitals Microapproach Disposable Femoral or Radial Vascular Access Introducer Sheath
Microapproach Disposable Femoral or Radial Vascular Access Introducer Sheath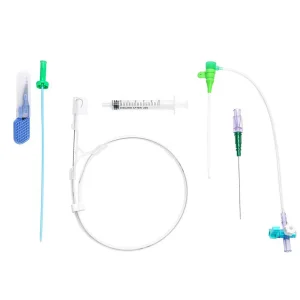 Premium Vascular Access Catheter Sheath for Reliable Hospital Use
Premium Vascular Access Catheter Sheath for Reliable Hospital UseThe global Transseptal Access System market is experiencing a paradigm shift driven by the rising incidence of Atrial Fibrillation (AFib) and the expansion of Left Atrial Appendage Closure (LAAC) procedures.
In today's interventional cardiology landscape, transseptal access serves as the "gateway" to the left heart. As structural heart interventions grow more complex, the demand for integrated systems that combine puncture needles, dilators, and steerable guiding sheaths has surged. This industrial evolution is moving away from generic tools toward specialized, procedure-specific systems designed for mitral valve repair, ablation, and paravalvular leak closure.
Current industrial trends emphasize miniaturization and radiopacity. Hospitals in North America and Europe are increasingly seeking "One-Shot" solutions—systems that provide stable access with minimal trauma, reducing procedural time and improving patient safety outcomes. As a result, the market is no longer just about the device; it's about the procedural efficiency it enables.

Modern systems now incorporate multi-directional steering mechanisms, allowing surgeons to navigate difficult anatomical variations in the right atrium with unprecedented precision.
The integration of Echo-compatible materials ensures that the transseptal needle and sheath are clearly visible under ICE (Intracardiac Echocardiography), reducing fluoroscopy time.
Next-generation dilators utilize hydrophilic coatings and tapered tips to ensure a smooth crossing of the septum, significantly lowering the risk of unintended perforations.

Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Through continuous innovation and strong R&D capabilities, SJ Medical has obtained multiple patents and international certifications, including CE, ISO 13485, and CFDA approval. We operate a modern 5,000 square meter facility with high-standard cleanrooms, ensuring every Transseptal Access System meets stringent global medical standards.
Why international hospitals and distributors are turning to China's leading manufacturers for Transseptal Access solutions.
We provide high-performance devices at a competitive price point, helping healthcare systems manage rising costs without compromising on patient clinical outcomes.
Our integrated manufacturing ensures consistent supply and rapid turnaround times, a critical factor for global procurement managers facing logistics challenges.
Leveraging our decades of engineering experience, we offer tailored solutions to meet specific regional clinical requirements and localized application scenarios.
As a leading China Transseptal Access System Manufacturer, SJ Medical bridges the gap between high-tech innovation and accessible healthcare. Our products are widely applied in minimally invasive procedures involving both coronary and peripheral vascular systems. They play an important role in the diagnosis and treatment of vascular diseases, helping to improve procedural efficiency while enhancing patient safety and recovery.
Our systems are engineered for diverse clinical environments, from state-of-the-art hybrid ORs in Europe to high-volume catheterization labs in Southeast Asia. Key applications include:
Currently distributed in: Italy, Finland, Spain, France, Netherlands, India, Indonesia, Turkey, Egypt, and beyond.
The company is equipped with advanced production and inspection systems, including precision balloon forming equipment, laser welding and marking technologies, and comprehensive laboratory testing facilities. These capabilities ensure high levels of manufacturing accuracy, product consistency, and full-process quality control.
Our million-class clean workshop and localized hundred-class areas for critical processes guarantee that every catheter and sheath is free from contaminants, ensuring bio-compatibility and patient safety during every invasive procedure.
Explore our full portfolio of vascular and renal access solutions.
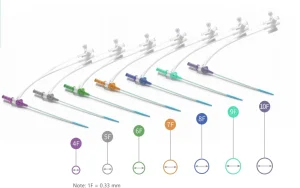 Microapproach CE Disposable Femoral Radial Vascular Access Introducer Sheath Kit Guiding Sheath
Microapproach CE Disposable Femoral Radial Vascular Access Introducer Sheath Kit Guiding Sheath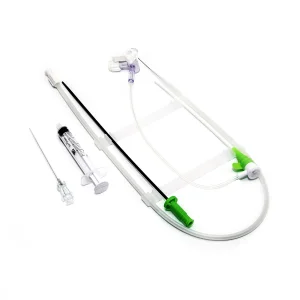 Microapproach Disposable Femoral or Trans Radial Vascular Access Introducer Sheath
Microapproach Disposable Femoral or Trans Radial Vascular Access Introducer Sheath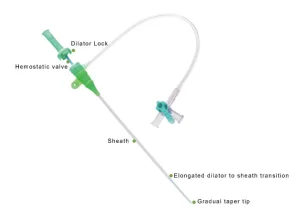 Premium Disposable Catheter Sheath for Enhanced Vascular Access
Premium Disposable Catheter Sheath for Enhanced Vascular Access Premium Vascular Access Catheter Sheath for Medical Professionals
Premium Vascular Access Catheter Sheath for Medical Professionals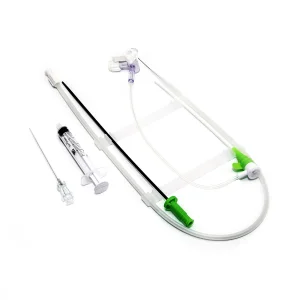 Femoral or Trans Radial Vascular Access Introducer Sheath
Femoral or Trans Radial Vascular Access Introducer Sheath Disposable Dilator and Peel-Away Sheath Medical Consumable for Efficient Vascular Access
Disposable Dilator and Peel-Away Sheath Medical Consumable for Efficient Vascular Access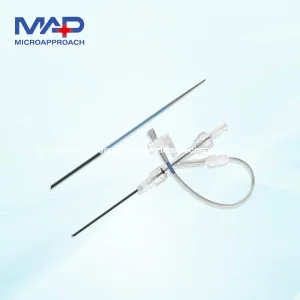 . Vascular Guiding Access Sheath for Intervention Surgery
. Vascular Guiding Access Sheath for Intervention Surgery Biocompatible Renal Ureteral Access Sheath
Biocompatible Renal Ureteral Access Sheath