 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
Innovative Balloon Dilatation Catheters for Critical Vascular Procedures
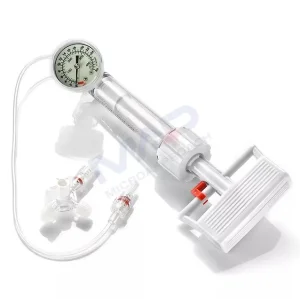 Balloon Expansion Pressure Pump Inflation Device PTCA Surgery
Balloon Expansion Pressure Pump Inflation Device PTCA Surgery
 Dedicated Vascular Balloon Catheter Chocolate Balloon
Dedicated Vascular Balloon Catheter Chocolate Balloon
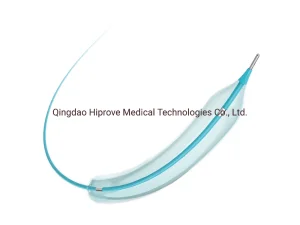 Interventional PTCA Balloon Dilatation Catheters
Interventional PTCA Balloon Dilatation Catheters
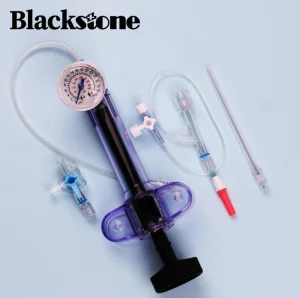 Renal Artery Angioplasty Balloon Inflation Device
Renal Artery Angioplasty Balloon Inflation Device
 Advanced Angioplasty Balloon for Vascular Access
Advanced Angioplasty Balloon for Vascular Access
 NC High Pressure Balloon Dilatation Catheter
NC High Pressure Balloon Dilatation Catheter
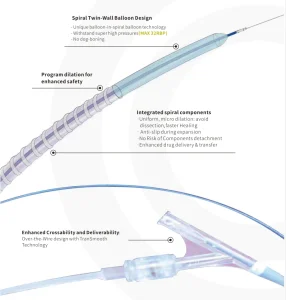 Durable PTA Spiral Balloon Catheter Procedures
Durable PTA Spiral Balloon Catheter Procedures
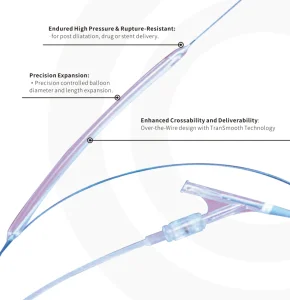 Precision Engineered PTA Balloon Dilatation
Precision Engineered PTA Balloon Dilatation
The global medical landscape is witnessing a paradigm shift from traditional open-heart surgeries to minimally invasive interventional procedures. At the heart of this revolution is the Valvuloplasty Balloon, a critical tool in treating valvular heart disease. As an industry-leading manufacturer, we recognize that the prevalence of aortic stenosis and mitral valve diseases, driven by an aging global population, has exponentially increased the demand for high-performance valvuloplasty systems.
The interventional cardiology market is projected to grow at a CAGR of 7.5%, with balloon valvuloplasty serving as a primary bridge to TAVI/TAVR procedures globally.
Modern trends focus on Ultra-High Pressure and Non-Compliant (NC) materials that offer precise expansion without causing annular rupture.
Global hospitals and GPOs are increasingly seeking Custom OEM partners who can provide tailored balloon lengths, diameters, and shaft stiffness for complex anatomies.
In regions like Europe and North America, the focus is on procedural safety and clinical efficiency. Meanwhile, in emerging markets across Asia and the Middle East, there is a rising need for high-quality yet cost-effective medical devices. As a premier China-based factory, Hangzhou SJ Medical Co., Ltd. bridges this gap by offering world-class engineering at competitive scales.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
The company is supported by a highly experienced technical team, many of whom have been engaged in the medical device industry for decades. Under the leadership of senior engineers, SJ Medical maintains strict quality control and continuously improves its manufacturing processes to ensure product reliability and consistency.
Our valvuloplasty balloon factories utilize laser welding and high-precision balloon forming machines to achieve ultra-thin profiles with high burst pressure resistance.
Located in Hangzhou's medical tech hub, we leverage a robust supply chain to ensure rapid prototyping and shortened lead times for large-scale OEM orders.
We don't just manufacture; we ensure global trust. Our products meet CE, ISO, and CFDA standards, facilitating smooth market entry for our international partners.
As a specialized Valvuloplasty Balloon manufacturer, we offer deep customization options:
SJ Medical focuses on the development of vascular interventional devices designed for both coronary and peripheral procedures. Its product range integrates interventional access systems, diagnostic catheters, and therapeutic devices, forming a complete solution for minimally invasive vascular treatments. Each product is developed with an emphasis on precision, safety, and clinical performance, aiming to support physicians in achieving optimal procedural outcomes.
SJ Medical strictly adheres to international quality standards and regulatory requirements. The company has obtained CE certification, ISO 13485 quality management system certification, and CFDA approval, along with free sale certificates, demonstrating its strong commitment to product quality and compliance.
The company’s products are widely applied in minimally invasive procedures involving both coronary and peripheral vascular systems. They play an important role in the diagnosis and treatment of vascular diseases, helping to improve procedural efficiency while enhancing patient safety and recovery.
The company is equipped with advanced production and inspection systems, including precision balloon forming equipment, laser welding and marking technologies, and comprehensive laboratory testing facilities. These capabilities ensure high levels of manufacturing accuracy, product consistency, and full-process quality control.
SJ Medical has established long-term partnerships with clients across Europe, Asia, the Middle East, and Africa. Its products are currently distributed in countries such as Finland, Italy, Spain, France, the Netherlands, India, Indonesia, Turkey, and Egypt, among others. The company continues to expand its international footprint and strengthen its global presence.
SJ Medical is committed to providing comprehensive and professional services throughout the entire customer journey. From pre-sales consultation and technical support to efficient order processing and responsive after-sales service, the company ensures that every customer receives timely assistance and reliable solutions.

The future of Valvuloplasty Balloon manufacturing lies in the intersection of digital technology and material science. We are currently exploring AI-driven simulation for balloon behavior under high-pressure scenarios, ensuring that every custom OEM design performs flawlessly in the clinical setting. Our R&D team is also investigating drug-eluting balloon (DEB) coatings that could potentially reduce restenosis following valvular dilation.
Furthermore, the trend toward ambulatory surgical centers (ASCs) means that balloons must be easier to use and compatible with smaller sheath sizes (Low-Profile designs). Our 2024 product roadmap includes the launch of ultra-low profile catheters that allow for faster hemostasis and quicker patient recovery. By choosing SJ Medical, you are not just choosing a factory; you are choosing a partner at the forefront of interventional cardiology innovation.
Precision-Crafted Interventional Devices for Global Medical Excellence
 Premium High Pressure Balloon Dilatation Catheter
Premium High Pressure Balloon Dilatation Catheter
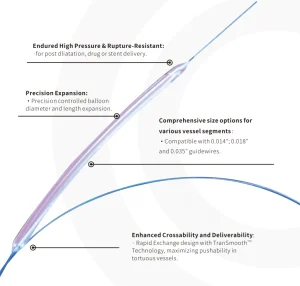 Reliable PTA Balloon for Patient Outcomes
Reliable PTA Balloon for Patient Outcomes
 2024 Balloon Catheter Inflation Device Manufacturer
2024 Balloon Catheter Inflation Device Manufacturer
 Medical PTCA Balloon Dilatation Catheter
Medical PTCA Balloon Dilatation Catheter
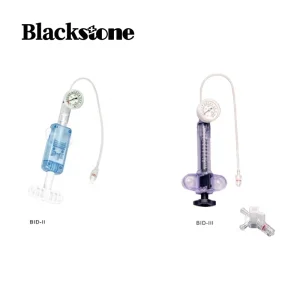 One Handed Prep PTCA Balloon Inflation Device
One Handed Prep PTCA Balloon Inflation Device
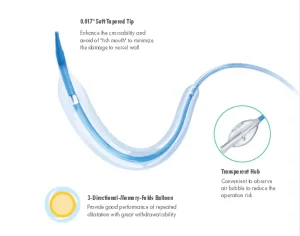 Disposable PTCA Balloon Dilatation Catheter
Disposable PTCA Balloon Dilatation Catheter
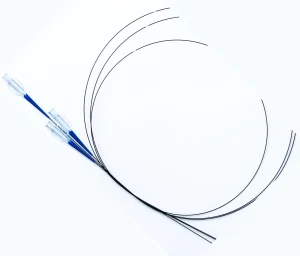 Customized Distal Finished Hypotube Intervention
Customized Distal Finished Hypotube Intervention
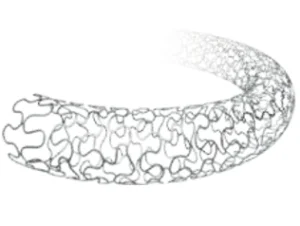 Polymer-Free Sirolimus-Eluting Coronary Stent System
Polymer-Free Sirolimus-Eluting Coronary Stent System