 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
 En 12662 Total Contamination Test Device for Middle Distillates
En 12662 Total Contamination Test Device for Middle Distillates
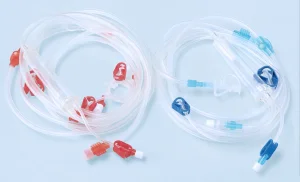 Durable Medical Blood Vessel Tubes for Hemodialysis Treatment
Durable Medical Blood Vessel Tubes for Hemodialysis Treatment
 Hemodialysis Machine in Nephrology Dept
Hemodialysis Machine in Nephrology Dept
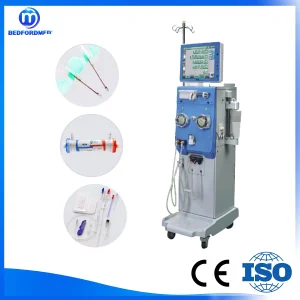 Medical Hemodialysis Machine Kidney Dialysis Equipment with CE
Medical Hemodialysis Machine Kidney Dialysis Equipment with CE
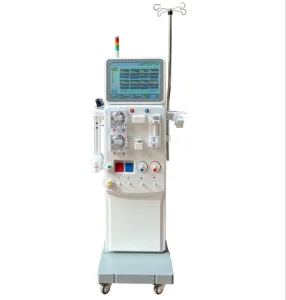 Hemodialysis Machines Are Used for Hemodialysis of The Kidneys
Hemodialysis Machines Are Used for Hemodialysis of The Kidneys
 Biobase Histology Tissue Embedding Center with LCD Display for Lab and Medical
Biobase Histology Tissue Embedding Center with LCD Display for Lab and Medical
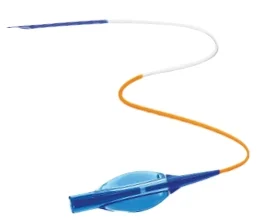 Advanced Thrombus Extraction Catheter for Efficient Blood Clot Removal
Advanced Thrombus Extraction Catheter for Efficient Blood Clot Removal
 Scaffold Clamp Adjustable Scaffold Coupler for Versatile Applications
Scaffold Clamp Adjustable Scaffold Coupler for Versatile Applications
In the high-stakes world of vascular health, the Vena Cava Filter (VCF) stands as a critical guardian against Pulmonary Embolism (PE). As a premier China Vena Cava Filter Factory, we recognize the shifting paradigms in global healthcare. The VCF market is no longer just about permanent implantation; it has evolved into a sophisticated field of retrievable designs and bio-compatible materials.
Pulmonary Embolism remains a leading cause of preventable hospital deaths worldwide. With the global aging population increasing, the demand for effective venous thromboembolism (VTE) management tools has reached an all-time high. Our role as a leading exporter is to bridge the gap between high-end medical technology and accessible healthcare solutions.

With decades of combined experience in Nitinol manufacturing and interventional design, our engineering team ensures every filter meets rigorous anatomical requirements.
As an ISO 13485 certified facility, our production protocols align with the highest international standards for class III medical devices.
Global clinicians trust our devices for their predictable deployment and high filtration efficiency, backed by extensive clinical data and post-market surveillance.
The industry is moving toward "Smart Intervention." Modern Vena Cava Filters are now focusing on:
Utilization of Nickel-Titanium (Nitinol) shape-memory alloys for better kink resistance and self-centering capabilities.
Enhanced retrieval hooks and structural stability allow for longer implantation windows before safe removal.
Low-profile delivery systems (5F-6F sheaths) that minimize vessel trauma at the access site.
For global procurement officers, these trends mean that selecting a Vena Cava Filter exporter requires looking beyond the price point. One must evaluate the manufacturer’s ability to innovate within these specific technological domains.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
The company is equipped with advanced production and inspection systems, including precision balloon forming equipment, laser welding and marking technologies, and comprehensive laboratory testing facilities. These capabilities ensure high levels of manufacturing accuracy, product consistency, and full-process quality control.
Why do international distributors choose us as their China Vena Cava Filter supplier? It’s the balance of cost-efficiency and uncompromising quality. Global procurement needs vary significantly by region:
Demand focus: CE compliance, clinical efficacy data, and sustainable packaging. Hospitals in Italy, France, and Spain require high-performance retrieval features.
Demand focus: Versatility in sizing to accommodate diverse patient anatomies and competitive pricing for tender-based public health systems.
Demand focus: Robust training and technical support, reliable supply chain stability, and ease of use in diverse clinical settings.
Our Vena Cava Filters are applied in various clinical scenarios including:
SJ Medical strictly adheres to international quality standards and regulatory requirements. The company has obtained CE certification, ISO 13485 quality management system certification, and CFDA approval, along with free sale certificates, demonstrating its strong commitment to product quality and compliance.
Our products are currently distributed in countries such as Finland, Italy, Spain, France, the Netherlands, India, Indonesia, Turkey, and Egypt. We offer comprehensive and professional services throughout the entire customer journey—from pre-sales consultation to responsive after-sales service.
 Versatile Steel Scaffold with Foldable Design and Wheels
Versatile Steel Scaffold with Foldable Design and Wheels
 Interventional Materials Balloon Dilatation Catheters Angioplasty Stent Ptca balloon Catheter
Interventional Materials Balloon Dilatation Catheters Angioplasty Stent Ptca balloon Catheter
 Low Level Laser Therapy Device for Chronic Acute Joint Pain
Low Level Laser Therapy Device for Chronic Acute Joint Pain
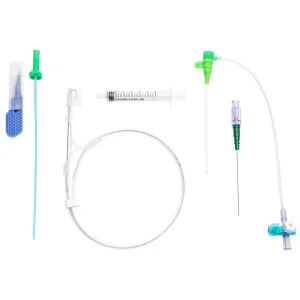
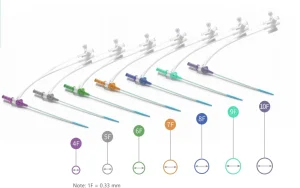 Microapproach CE Disposable Femoral Radial Vascular Access Introducer Sheath Kit Guiding Sheath
Microapproach CE Disposable Femoral Radial Vascular Access Introducer Sheath Kit Guiding Sheath
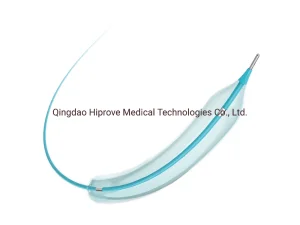
 Premium Surgical Catheter Sheath for Optimal Medical Procedures
Premium Surgical Catheter Sheath for Optimal Medical Procedures
 Durable Ringlock Scaffold with Anti Corrosion and Anti Oxidation Protection
Durable Ringlock Scaffold with Anti Corrosion and Anti Oxidation Protection
 Biobase Medical Pathology Tissue Embedding Center
Biobase Medical Pathology Tissue Embedding Center
 Below Knee Prosthetic Components for Prosthetics Leg
Below Knee Prosthetic Components for Prosthetics Leg
The rise of China as a hub for medical device manufacturing, particularly in the interventional vascular space, is driven by a unique confluence of factors. First, the industrial cluster effect in cities like Hangzhou provides unparalleled access to raw materials and specialized components like high-grade Nitinol wire and medical-grade polymers. This ecosystem allows a Vena Cava Filter factory to rapidly prototype and iterate designs based on feedback from top-tier domestic hospitals.
Second, the investment in R&D and Human Capital. Our technical team consists of veterans who have seen the evolution of vascular surgery from open procedures to minimally invasive techniques. This deep-rooted expertise translates into devices that are not just functional but "physician-friendly." We understand the nuances of the "feel" of a catheter as it navigates the iliac vein and the importance of radiopacity in the filter struts.
Third, Supply Chain Resilience. In a post-pandemic world, global procurement officers prioritize reliability. By maintaining a 5,000sqm facility with integrated manufacturing and testing, we control the entire lifecycle of the product. This reduces lead times and ensures that our global partners in Europe and Asia are never out of stock.
Fourth, the Digital Transformation. We utilize AI-enhanced quality inspection systems to detect microscopic defects in stent and filter structures that the human eye might miss. This "Tech-Forward" approach is what defines a modern China exporter. Our commitment to the E-E-A-T principle means we don't just sell products; we offer documented reliability and clinical peace of mind.
In conclusion, whether you are a hospital procurement manager in Finland or a medical device distributor in India, partnering with a leading China Vena Cava Filter exporter like SJ Medical provides you with a competitive edge. You gain access to world-class innovation, certified quality, and a dedicated team committed to advancing vascular health globally.