 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
Providing Advanced Vascular Interventional Solutions for the Nordic Medical Market
Norway represents one of the most sophisticated healthcare markets in Europe, driven by a robust public health system and a commitment to high-technology medical interventions. As a leading Atherectomy Systems Factory & Supplier, we recognize that the Norwegian medical landscape is increasingly focused on minimally invasive techniques to treat Peripheral Artery Disease (PAD) and Coronary Artery Disease (CAD). In major medical hubs like Oslo, Bergen, and Trondheim, clinical excellence is the standard, requiring suppliers to demonstrate not only technical proficiency but also a deep understanding of Nordic healthcare regulations.
The demand for advanced atherectomy technology in Norway is fueled by an aging population and a high prevalence of calcified vascular lesions. Traditional angioplasty often faces challenges in these complex cases, leading to a surge in the adoption of rotational, orbital, and directional atherectomy systems. Our factory specializes in producing the precision components and integrated systems that allow Norwegian surgeons to achieve optimal vessel preparation before stenting or drug-coated balloon application.
Modern atherectomy systems are moving toward integration with Intravascular Ultrasound (IVUS) and Optical Coherence Tomography (OCT). This allows Norwegian clinicians to visualize plaque morphology in real-time, ensuring precise excision without damaging the vessel wall.
In alignment with Norway's "Green Shift" (Grønt skifte), our factory is implementing sustainable manufacturing processes, reducing carbon footprints in the production of medical-grade catheters and wires.
The trend is shifting toward "vessel-specific" tools. Our R&D team works on customizable catheter lengths and burr sizes to cater to the specific anatomical needs of the Nordic demographic.
As a supplier to the Norwegian market, we adhere to the strict E-E-A-T principles. Our Experience is reflected in over a decade of partnership with global cardiology leaders. Our Expertise is proven by our million-class clean workshops and precision laser welding technologies. Our Authoritativeness is established through CE and CFDA certifications, and our Reliability is seen in our consistent product performance in high-pressure clinical environments.
Hangzhou SJ Medical Co., Ltd. was established in 2015 and is dedicated to the research, development, manufacturing, and commercialization of vascular interventional medical devices. The company focuses on both cardiovascular and peripheral intervention fields, striving to provide safe, reliable, and innovative solutions for healthcare providers worldwide.
Over the years, SJ Medical has developed a comprehensive product portfolio covering key areas of vascular intervention. Its offerings span from various types of balloon catheters for coronary and peripheral applications to supporting access and diagnostic devices, as well as advanced stent systems. Through continuous innovation and strong R&D capabilities, the company has obtained multiple patents and international certifications, laying a solid foundation for sustainable growth in both domestic and global markets.

SJ Medical operates a modern manufacturing facility with a total area of approximately 5,000 square meters. The factory is built in accordance with GMP standards and features a high-standard cleanroom environment, including a million-class clean workshop with localized hundred-class areas for critical processes.
The company is supported by a highly experienced technical team, many of whom have been engaged in the medical device industry for decades. Under the leadership of senior engineers, SJ Medical maintains strict quality control and continuously improves its manufacturing processes to ensure product reliability and consistency.
Our products are tailored for specific clinical scenarios encountered in Norwegian hospitals:
SJ Medical strictly adheres to international quality standards and regulatory requirements. The company has obtained CE certification, ISO 13485 quality management system certification, and CFDA approval, along with free sale certificates, demonstrating its strong commitment to product quality and compliance.
Our global footprint includes long-term partnerships in Finland, Italy, Spain, France, the Netherlands, and now expanding rapidly within Norway to support the local vascular surgery community. We provide comprehensive services from pre-sales consultation to responsive after-sales support.

 Premium Surgical-Grade Introducer Catheter Sheath for Professionals
Premium Surgical-Grade Introducer Catheter Sheath for Professionals
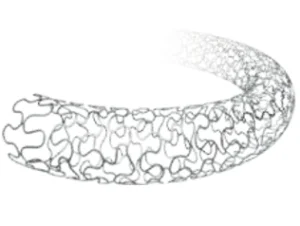 Polymer-Free Sirolimus-Eluting Coronary Stent System - Norway Standard
Polymer-Free Sirolimus-Eluting Coronary Stent System - Norway Standard
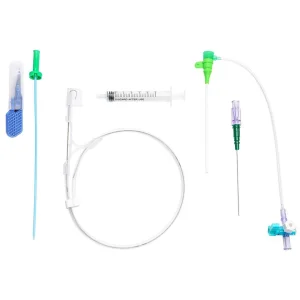 Premium Catheter Sheath for Cardiology - Single-Use Medical Supply
Premium Catheter Sheath for Cardiology - Single-Use Medical Supply
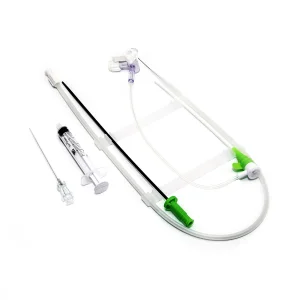 Femoral or Trans Radial Vascular Access Introducer Sheath
Femoral or Trans Radial Vascular Access Introducer Sheath
 Vascular Sheaths for Femoral/Radial Artery (4/5/6/7/8 French)
Vascular Sheaths for Femoral/Radial Artery (4/5/6/7/8 French)
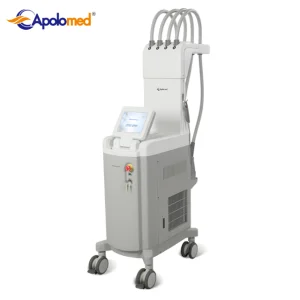 1060nm Laser Diode Device for Advanced Vascular Therapy
1060nm Laser Diode Device for Advanced Vascular Therapy
 Customed Prosthetic Socket Leg for Nordic Rehabilitation
Customed Prosthetic Socket Leg for Nordic Rehabilitation
 High Pressure PTCA Balloon Catheter for Coronary Heart Disease
High Pressure PTCA Balloon Catheter for Coronary Heart Disease
Looking for a reliable partner in Norway? Our technical experts are ready to assist with your vascular intervention needs.
Connect with Our Norway Team