 SJ Medical
SJ Medical
 SJ Medical
SJ Medical
Houston, Texas, is home to the world-renowned Texas Medical Center (TMC), the largest medical complex on the planet. As a premier Renal Artery Denervation Factory & Exporter in Houston, we leverage this unique ecosystem of research hospitals, biotechnology startups, and clinical trial expertise. The city's industrial landscape has transitioned from energy dominance to a life-sciences powerhouse, creating a fertile ground for developing sophisticated vascular interventional devices.
Our presence in Houston allows us to collaborate with leading nephrologists and cardiologists to refine Renal Artery Denervation (RDN) technologies. RDN is a transformative, minimally invasive procedure targeting resistant hypertension by ablating the sympathetic nerves in the renal arteries. By manufacturing locally and exporting globally, we bridge the gap between cutting-edge American clinical research and affordable, high-quality medical device distribution.

The global medical community is witnessing a massive shift toward device-based hypertension management. Recent clinical trials (such as SPYRAL HTN and RADIANCE) have solidified the efficacy of RDN, leading to a surge in demand for specialized catheters and access systems. As an industry leader, SJ Medical focuses on the latest trends: radiofrequency (RF) ablation and ultrasound-based denervation.
In Houston’s high-tech corridors, the trend is moving toward "Smart Catheters" integrated with AI diagnostics. Our R&D focuses on multi-electrode systems that ensure circumferential ablation while preserving the integrity of the arterial wall—precision that is essential for long-term patient safety and procedural success.
Hangzhou SJ Medical Co., Ltd., established in 2015, is a specialized manufacturer dedicated to the research, development, and commercialization of vascular interventional devices. We bridge the technology gap between East and West, operating as a vital exporter for the Houston medical tech corridor.
Operating a 5,000 sqm modern facility built to GMP standards, our manufacturing environment includes million-class cleanrooms. We utilize precision laser welding and advanced balloon forming technology to ensure every catheter meets the rigorous standards required by Houston’s top-tier hospitals.
From Houston to the Middle East, and from Europe to Asia, our products are trusted in over 20 countries. We maintain long-term partnerships in Finland, Italy, India, and Turkey, providing the logistical reliability expected of a top-tier exporter.

In Houston's clinical setting, Renal Artery Denervation is primarily utilized in Catheterization Laboratories (Cath Labs). Our specialized access sheaths and dilators are essential components for the following scenarios:
Get in touch for bulk orders, custom OEM manufacturing, or technical specifications of our Renal Artery Denervation systems.
Send Inquiry Now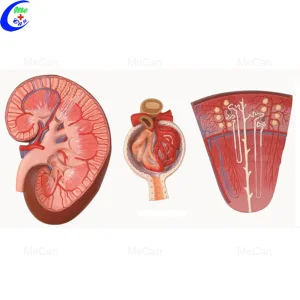 Kidney with Adrenal Model: Houston Clinical Education Tool
Kidney with Adrenal Model: Houston Clinical Education Tool
 Houston Precision Renal Dilator Set with Sheath & Guide Wire
Houston Precision Renal Dilator Set with Sheath & Guide Wire
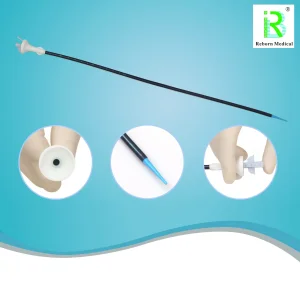 Biocompatible Renal Ureteral Access Sheath - Texas Series
Biocompatible Renal Ureteral Access Sheath - Texas Series
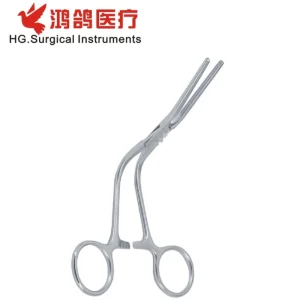 Houston Medical Grade Renal Vascular Forceps
Houston Medical Grade Renal Vascular Forceps
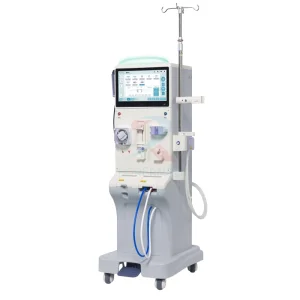 Houston High-Flow Hemodialysis Kidney Machine
Houston High-Flow Hemodialysis Kidney Machine
 High Accuracy Houston-Spec Renal Function Meter
High Accuracy Houston-Spec Renal Function Meter
 Ureteral Stents: Structural Support for Houston Nephrology
Ureteral Stents: Structural Support for Houston Nephrology
 Nephrology Department Hemodialysis Machine - Houston Grade
Nephrology Department Hemodialysis Machine - Houston Grade
In the medical device industry, reliability is non-negotiable. As a prominent Renal Artery Denervation Factory & Exporter, SJ Medical adheres to the strictest international protocols. We have secured CE Certification, ISO 13485, and CFDA Approval, ensuring our devices are ready for use in the highly regulated markets of Houston and the wider United States.
Our commitment to Quality Control (QC) involves rigorous testing of balloon burst pressures, fatigue testing of catheters, and biocompatibility assessments of all materials. In Houston, where clinical excellence is the benchmark, our products provide the consistency required by world-class surgeons.
We provide more than just hardware; we provide a complete partnership including:
The city's focus on "Medical Device Innovation" has attracted over 1,760 life sciences companies. Our export strategy specifically targets the Houston-The Woodlands-Sugar Land metropolitan area, providing essential components that fuel the local healthcare economy. By combining Houston's clinical insight with our advanced manufacturing capabilities, we create a synergy that benefits patients worldwide.